Omar Alzarkali
Batavia, New York, United States
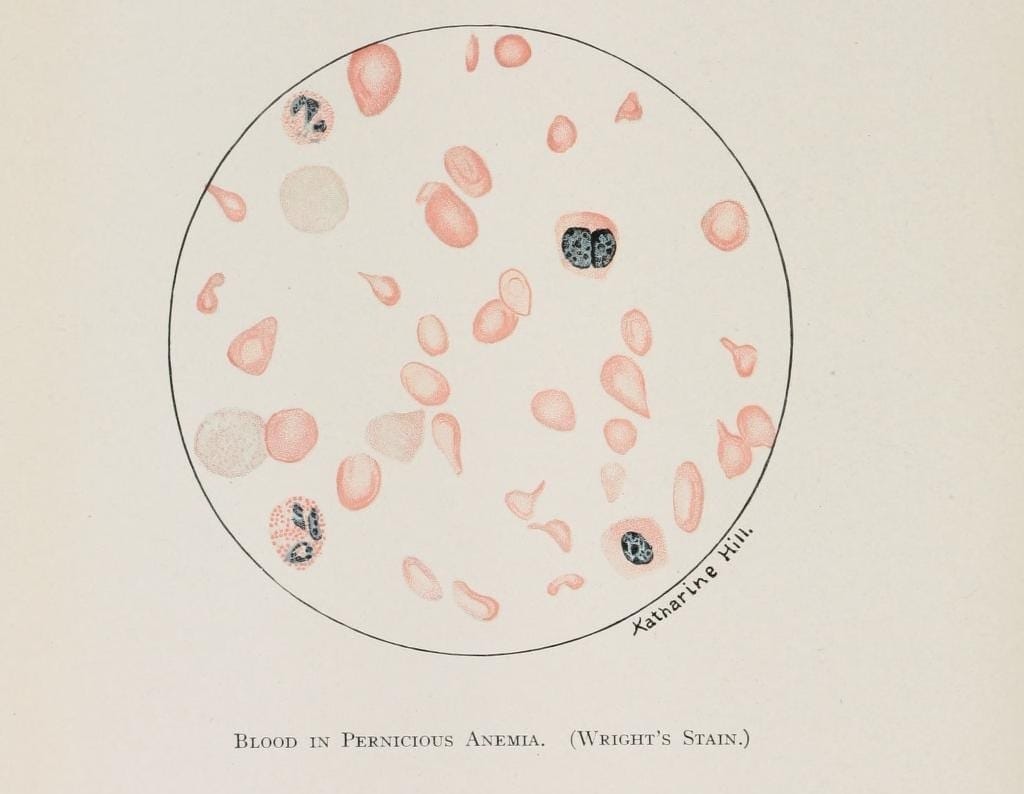
Blood is powerful. The mere sight of it can cause an adult to fall to the ground; as a medical student, I have seen it happen. Faces go pale and legs can no longer carry their weight as they succumb to this primitive reflex. Perhaps this vasovagal response happens because we know that blood belongs inside of us, sustaining us with the oxygen it carries and clears poisons from our cells. It tells us of our diseases in a language translated by technicians that guides healers to the appropriate antidote.
The liver is often noted as the most multi-faceted organ. It is burdened by yet resilient to the challenges we throw at it. Individually the liver and blood are fascinating, but the symbiotic relationship between them is even more marvelous. As the liver receives its supply of blood, it harvests the iron, folate, and vitamin B12 the blood needs to keep the rest of the body functioning well.
Our blood cells, like other cells in our body, are subject to old age, expenditure of their capabilities, and eventually death. They are destroyed at the end of approximately four months by the very oxygen they deliver in a process known as oxidative stress. The bone marrow has to ensure a constant supply of new cells to propagate this cycle. As bone marrow stem cells replicate to make new cells, they depend on raw materials to forge these replacements. Among these raw materials, cobalamin, more commonly known as vitamin B12, is essential for the synthesis of new DNA. Lack of this vitamin leads to the formation of large, immature, and inefficient red blood cells that cause the eventual collapse of the entire system. The importance of this nutrient is evident in the liver’s ability to store a supply to last for years.
Humans need both an adequate dietary supply of B12 and a binding protein produced by the stomach, intrinsic factor, to facilitate its absorption.1 Intrinsic factor was discovered by William B. Castle, who coined the term in 1929. The term intrinsic factor at first glance seems like an unusually vague name for something with such a unique function. Like the terminology of many scientific discoveries, a close examination of the historical etymology helps us understand why it was given this name. He appreciated its relationship with the then unknown substance that prevented pernicious anemia, which he named “extrinsic factor” and is now known as vitamin B12.2 In 1934, the Nobel Prize for Physiology and Medicine was awarded to George Hoyt Whipple, George Richards Minot, and William Parry Murphy for work based on the initial work of Castle and for discovering the cure for pernicious anemia.3
Minot was fascinated by blood and believed in the relationship between good food and good health. Having been diagnosed as having severe diabetes one year before the discovery of insulin, Minot “ate no food at home that was not weighed or measured and recorded. When dining out, advance knowledge of the menu aided him in estimating calories and carbohydrates.”4 His passion drove him to research hematological diseases and his traditional values made him aware of his patients’ diets. The relationship between these two forces put him on the path to unlocking the secret of pernicious anemia. Noticing a pattern of poor diets among his patients afflicted with the disease, he partnered with Murphy and they began to experiment with diets based on earlier work of Whipple with anemic dogs. Whipple had noticed that blood synthesis greatly improved in these dogs with certain foods. Experimenting with various diets and quantities, they noticed that feeding their patients animal liver had the most drastic results, entirely reversing their condition and effectively curing them of their once terminal and mysterious illness.4
In an effort to further their knowledge of the disease, Minot and Murphy sought the help of Edwin Joseph Cohn, a professor of physical chemistry at Harvard Medical School, to isolate the specific molecule in liver that cured pernicious anemia. This led to the discovery of what they came to call “water soluble vitamin B” as the agent responsible for curing the disease.5 While eating a large quantity of liver daily was an effective treatment, it was not convenient. By eliminating the nonessential components of liver, the delivery of the cure became more tolerable.4
The relationship between the blood, liver, bone marrow, and gut represents a whole that surpasses the sum of its parts. In a similarly collaborative manner, the discovery of the physiology, pathology, cure, and overall knowledge of pernicious anemia is a testament to what is achievable through the combined efforts of many dedicated scientists. This once fatal disease is now easily treatable and preventable thanks to no individual pioneer but to the collective work of a passionate community whose members built on the parts of their predecessors.
References
- Pocock G, Richards C (2006). Human Physiology: The Basis of Medicine (3rd ed.). Oxford University Press. p. 230. ISBN 978-019-856878-0.
- Greer JP (2014). Wintrobe’s Clinical Hematology Thirteenth Edition. Philadelphia, PA: Wolters Kluwer/Lippincott Williams & Wilkins. ISBN 978-1-4511-7268-3. Chapter 36: Megaloblastic anemias: disorders of impaired DNA synthesis by Ralph Carmel.
- The Nobel Prize in Physiology or Medicine 1934. NobelPrize.org. Nobel Media AB 2019. Sat. 14 Sep 2019. <https://www.nobelprize.org/prizes/medicine/1934/summary/>.
- Staff, N. and Secretary, O. (1974). Biographical Memoirs. Washington: National Academies Press, pp.337-383.
- Harris J. George R. Minot helped cure pernicious anemia. Healio.com. https://www.healio.com/hematology-oncology/news/print/hemonc-today/%7Be12289ef-171e-4231-a8be-a6b937cf5b02%7D/george-r-minot-helped-cure-pernicious-anemia. Published 2008. Accessed January 5, 2020.
OMAR ALZARKALI was raised in Syria by parents who emphasized education as the path to freedom. After high school, he moved to Orlando and graduated with a Bachelor of Science in Biomedical Sciences. He received a Masters in Medical Science from Loyola University Chicago and is currently a medical student at Lake Erie College of Osteopathic Medicine.
Submitted for the 2019–2020 Blood Writing Contest
