Jayant Radhakrishnan
Darien, Illinois, United States
Anant Radhakrishnan
Amarillo, Texas, United States

The term cloaca was first used around 600 BC by the Romans who named their main drainage channel the “Cloaca Maxima” or the Greatest Sewer. It drained the local marshes and all water and effluent from Rome into the Tiber River. They continued expanding it so that all eleven Roman aqueducts drained into it by the first century AD. According to them, the Cloaca Maxima was presided over by the goddess Cloācīna, or “the Purifier” (cf. Latin cluere = purify).1
Like its namesake in Rome, the cloaca in animals is a storehouse and conduit for excretory products. It is the only orifice at the rear end of many vertebrate animals for the urinary and genital tracts and the hind gut to communicate with the exterior. Invertebrates do not have a cloaca, although the tenia solium has a contractile caudal vesicle and the paramecium has two such vesicles, one at each end of the alimentary tract.2
Among vertebrates, all birds, reptiles, and amphibians have cloacae. The only mammals to have a true cloaca seem to have evolved in isolation away from their parent groups many millennia ago. Some examples of this convergent evolution are marsupial moles of Australia, golden moles of Southern Africa, and tenrecs of Madagascar and monotremes (Greek monós = single, trêma = hole), which are similar to the platypus that lays eggs instead of delivering live offspring.3
As mentioned above, the main function of the cloaca is to store and to evacuate urine and stool when required. But it is also a reproductive organ and has a role in thermoregulation and respiration under extreme circumstances.
While some birds use a phallus for reproduction, others such as owls reproduce by touching their cloacae together just long enough to permit sperm to be transferred from the male to the female. This is known as the “cloacal kiss.”4 Reptiles also have a cloaca, but the males have either one penis (crocodiles, turtles) or two hemipenises (snakes and lizards) inside the male cloaca that are purely for reproduction and separate from the urinary tract.
The cloaca’s role in thermoregulation by evaporative water loss (EWL) was first hypothesized by DeNardo when he saw a Gila monster roaming around in the hot Sonoran Desert with a prolapsed cloaca. DeNardo and his associates devised a system to measure cutaneous, respiratory (from the oral mucous membrane), and cloacal water loss when the reptile was subjected to various ambient temperatures. They found that cloacal EWL rose dramatically above a temperature of 35°C and it had the ability to cool the reptile’s body temperature by 3°C. If the animal was dehydrated to 80% of its initial body mass, the process was delayed and the increase in EWL was much reduced. This experiment clearly demonstrated that the mucus membranes of the cloaca could be a source of EWL and that the water loss occurred through its vent even without the cloaca being everted outside the body.5 These researchers then wondered whether birds could also use the cloaca to cool their body temperature in dire circumstances. They studied Inca doves and Eurasian quail. In the doves, evaporation from the cloaca was negligible up to an ambient temperature of 40°C. When ambient temperatures were raised to 42°C, the cloaca accounted for 21.2% while cutaneous and buccopharyngeal EWL constituted 53.4% and 25.4% respectively of the total EWL. Thus, cloacal evaporation shed approximately 150mW of heat at this temperature. The quails, on the other hand, did not have the capacity to use the cloaca for thermoregulation. They tolerated a maximum ambient temperature of 32°C, at which point their cloacal EWL constituted only 6.4% of the total water lost by evaporation. Whether they would have been able to increase EWL and control their body temperature at higher ambient temperatures could not be evaluated as the birds became distressed and the experiment had to be terminated.6
Another extremely valuable role of the cloaca is in enteral respiration under dire circumstances. Some species use the cloaca to exchange gases when they are hypoxic. In 1960, Dunson7 demonstrated that the Gulf Coast soft-shelled turtle had the ability to use the cloaca for gas exchange, although it was not very efficient. Bucco-pharyngeal respiration was the most efficient and it accounted for almost the entire oxygen uptake of the turtle’s total respiration. Lüdicke had previously reported in 19368 that “aquatic turtles respire under water by bucco-pharyngeal movements forcing water into and out of the vascular mouth.” He also noted that “the cloacal bladder expands simultaneously with the lungs, and is concerned with underwater respiration”7 of turtles. One of Lüdicke’s turtles lived underwater for eight days. The ability to use the cloaca for respiration is of value when it is difficult to breathe, such as when the turtle cannot rise to the surface to breathe as in a frozen pond or in a river that is flowing too fast. An example is the Australian Fitzroy River turtle, which can potentially obtain all its oxygen needs by cloacal ventilation. It has also been shown that the pond loach burrows into the mud and breathes through its rear end if its habitat dries up.9
These findings made others wonder if enteral ventilation could be of value in mammals. Okabe et al10 demonstrated that hypoxic mice died in eleven minutes while those that were administered oxygen transrectally survived for eighteen minutes. If they improved gas exchange further by abrading the intestinal mucosa, the mice had a 75% survival at fifty minutes. The researchers question whether transrectal oxygenation with oxygen in gas form or with liquid ventilation using perfluorocarbon could have a role as an adjuvant to standard respiratory therapy in human beings.
A particularly remarkable double role played by the cloaca is in sea cucumbers, which have a pair of respiratory trees in the cloaca on either side of the digestive tract. They breathe by drawing in and expelling water through the anus. They have a symbiotic and commensal relationship with pearl fish, bristle worms, and crabs, which utilize the continuous flow of water in the cloacal respiratory tree of the sea cucumber to exchange gases while also being protected from predators by residing there. They exit the sea cucumber’s anus at night to find food.11
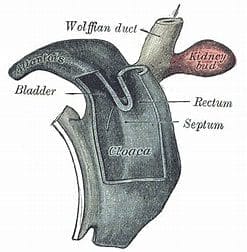
The majority of mammals have evolved away from having a cloaca at birth; however, all mammalian fetuses go through a stage of development when they have a cloaca that divides into an anterior genitourinary system and a posterior hind gut.12,13 The human fetus develops a cloaca by the fifth week of development, and normally by the seventh week, it divides into an anterior urogenital and posterior anorectal section because of differential growth between its ventral and craniodorsal parts.14 Sometimes, for unknown reasons, the cloaca does not divide and the baby is born with a “persistent” cloaca. This anomaly is believed to occur in approximately 1:20,000-50,000 births. It typically occurs in females, though a handful of boys have also been reported to have a cloaca. In humans a persistent cloaca must be corrected surgically as stasis of urine in the urinary bladder greatly increases the risk of urinary tract infections upon contamination with stool. Apart from complicated surgical corrections, the management of these children has to consider that they also have one or more associated anomalies that may affect the spine, kidneys, heart, esophagus, and limbs. With a lifetime of proper care, these patients can lead a full and productive life.
Since organisms adapt to their environment by developing traits that they pass on to their progeny for survival of the species, one has to assume that cloacal division gives mammals some advantage. Separation of the three tracts could conceivably improve storage, emptying, and continence of urine and stool. It could also possibly prevent cross-infection or benefit reproductive capabilities.
Bibliography
- de Vaan M (2008). Etymological Dictionary of Latin and the other Italic Languages. Boston, Leiden, p122. Brill. ISBN 9789004167971.
- Anjali S. Excretory organs in invertebrates. Zoology. Notes on Zoology.com https://www.notesonzoology.com/invertebrates/ accessed on September 22, 2023.
- Cloaca -Wikipedia. https://en.wikipedia.org/wiki/Cloaca accessed on September 22, 2023.
- Lynch W (2007). “The cloacal kiss” Owls of the United States and Canada. Baltimore, Johns Hopkins University Press, p151. ISBN 978-0-8018-8687-4.
- DeNardo DF, Zubal TE, Hoffman TCM (2004). Cloacal evaporative cooling: A previously undescribed means of increasing evaporative water loss at higher temperatures in a desert ectotherm, the Gila monster Heloderma suspectum. J Exp Biol. 207(6):945-953. https://doi.org/10.1242/jeb.00861.
- Hoffman TCM, Walsberg GE, DeNardo DF (2007). Cloacal evaporation: An important and previously undescribed mechanism for avian thermoregulation. J Exp Biol. 210(5):741-749. TE, https://doi.org/10.1242/jeb.02705Zubal.
- Dunson, WA (1960). “Aquatic Respiration in Trionyx spinifer asper”. Herpetologica 16(4):277–283. https://www.jstor.org/stable/3889486. JSTOR 3889486.
- Lüdicke M (1936). Uber die Atmung von Emys orbicularis. Zool Jahrb, Zool Physiol 56:83-106. Cited by Randall WC, Stullken DE, Hiestand WA (1944). In respiration of reptiles as influenced by the composition of the inspired air. Copeia 1944(3):136-144. https://doi.org/10.2307/1437805 https://www.jstor.org/stable/1437805.
- Ghosh S K, Ghosh B, Chakrabarti P (2011). Fine anatomical structures of the intestine in relation to respiratory function of air-breathing loach, Lepidocephalichthys guntea. Acta Ichthyologica et Piscatoria 41(1):1–5.
- Okabe R, Chen-Yoshikawa TF, Yoneyama Y, Yokoyama Y, Tanaka S, Yoshizawa A, et al (2021). Mammalian enteral ventilation ameliorates respiratory failure. Med 2:773-783. https://doi.org/10.1016/j.medj.2021.04.004.
- Toonen, R.J. (2003). Invertebrate Non-Column: Sea cucumbers – Part II. in Advanced Aquarists Online Magazine. January 2003. http://www.advancedaquarist.com/2003/1/inverts.
- van der Putte SCJ (1986). Normal and abnormal development of the anorectum. J Pediatr Surg. 21(5):434-440. https://doi.org/10.1016/S0022-3468(86)80515-2.
- Kluth D, Hillen M, Lambrecht W (1995). The principles of normal and abnormal hindgut development. J Pediatr Surg 30(8):1143-1147. doi: 10.1016/0022-3468(95)90007-1.
- Kruepunga N, Hikspoors JPJM, Mekonen HK, Mommen GMC, Meemon K, Weerachatyanakul W, et al (2018). The development of the cloaca in the human embryo. J Anat. 233:724-739. doi: 10.1111/joa.12882.
JAYANT RADHAKRISHNAN, MB, BS, MS (Surg), FACS, FAAP, completed a Pediatric Urology Fellowship at the Massachusetts General Hospital, Boston, following a Surgery Residency and Fellowship in Pediatric Surgery at the Cook County Hospital. He returned to the County Hospital and worked as an attending pediatric surgeon and served as the Chief of Pediatric Urology. Later he worked at the University of Illinois, Chicago, from where he retired as Professor of Surgery & Urology, and the Chief of Pediatric Surgery & Pediatric Urology. He has been an Emeritus Professor of Surgery and Urology at the University of Illinois since 2000.
ANANT RADHAKRISHNAN, DVM, ACVIM, graduated from the University of Georgia Veterinary School in Athens and then completed a residency in Small Animal Internal Medicine at the University of Pennsylvania in 2005. He then set up a small animal internal medicine practice in Lexington, Kentucky and developed it into a multispecialty hospital, the Blue Grass Veterinary Specialists and Animal Emergency. He is now Professor of Small Animal Internal medicine at the Texas Tech University School of Veterinary Medicine in Amarillo, Texas.
