George Dunea
Chicago, Illinois, United States
Twentieth century
Three major developments—renal biopsy, dialysis, and transplantation—revolutionized nephrology in the second half of the 20th century. Renal biopsy transformed the diagnostic approach to renal disease from a clinical methodology to one based on morphological analysis. Presently over one million patients with renal failure are maintained by dialysis throughout the world; the results of kidney transplantation are steadily improving; and spectacular advances in renal physiology and basic sciences promise to bring about even better therapeutic modalities. The general understanding of the pathogenesis and natural history of many renal diseases has also improved, resulting in changing nomenclatures and classifications of many clinical entities.
Glomerulonephritis
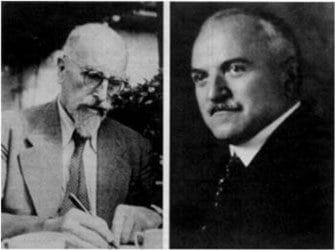
Franz Volhard (left) and Theodor Fahr (right)
The seminal observations of Richard Bright and his early successors were followed later by renewed efforts to understand and classify the various forms of glomerulonephritis. In 1905 Friedrich Mueller separated inflammatory from “degenerative” diseases by calling the latter “nephrosis.”1 A few years later F. Munk was so impressed by the fat bodies in the urine of nephrotic patients that he coined the term lipoid nephrosis.2 In the 1920s Henry Christian emphasized that “nephrosis” was a syndrome and mostly a manifestation of glomerulonephritis. But the most profound influence on the nosology of renal disease came from the publication in 1914 of Volhard and Fahr’s famous monograph “Die Brightsche Nierenkrankheit.”3–5
Based on clinical observations and on the autopsy material available at the time, these investigators formulated a classification that remained in use well into the age of renal biopsy, dividing diffuse glomerulonephritis into acute, subacute (extracapillary or crescentic), and chronic forms. Other attempts at classification defined subacute “hydremic” versus “parenchymatous” forms, and Dorothy Russell’s diphtheria based nomenclature of nephritis mitis, gravis, and intemedia. Between 1927 and 1936 Longcope divided nephritis into two broad groups, type A and B, essentially distinguishing nephritic from nephrotic cases. Arthur Ellis extended this concept in 1942 with an influential classification in which type I consisted largely of proliferative nephritic cases, while type II was characterized by the insidious appearance of nephrotic edema.3–6
Further advances in the pre-biopsy era included the description of membranous nephropathy by E. T. Bell. In the 1950s A. C. Allen published a beautifully illustrated atlas of renal diseases featuring, among other conditions, a lobular form now corresponding predominantly to membranoproliferative glomerulonephritis. Still in the pre-biopsy era, Thomas Addis recognized in his classical monograph “Glomerular Nephritis” three major forms of the disease: (i) hemorrhagic (acute, subacute, chronic), (ii) degenerative (nephroses and toxins) and (iii) arteriosclerotic; also commenting at the time that “all we know for certain about the kidney is that it makes urine.”3–5
Acute renal failure
Although acute suppression of urine and its ominous import had been recognized since antiquity, a better understanding of its natural history had to await the two more recent World Wars. Hackracht in 1917 wrote about the renal failure of German soldiers sustaining crush injuries in the trenches. Between the wars several reports of acute renal failure and shock kidney were concealed under different names in a mix of cases, so that their common basis escaped recognition.7,8 But in 1941 Eric Bywaters reported on the acute renal failure of crush injury sustained by victims of the blitz bombing of London, describing the clinical stages of onset, oliguria, polyuria, and eventual return of function, noting severely damaged renal tubules and myoglobinuric casts.8 He used the term “acute tubular necrosis,” which became the accepted designation of nonspecific acute renal failure, even though others favored vasomotor nephrosis or nephropathy.9
Lucke in 1946 concluded from anatomical studies that the renal injury was in the distal convoluted tubules, and coined the term “lower nephron nephrosis.” Further evidence of a tubular lesion was adduced by the classical microdissection studies of Jean Oliver, all this emphasizing the importance of tubular damage. In recent years the term acute renal injury has become increasingly used to denote non-specific ischemic or nephrotoxic renal failure, but the precise mechanism of its causation remains incompletely understood.9,10
Landmarks in the therapeutic approach to acute renal failure included a renewed emphasis on sound principles of conservative management by Graham Bull and his colleagues in the 1950s, and on prophylactic dialysis by Paul Teschan during the Korean War. Treatment of severe renal failure by dialysis is now standard practice, but mortality has remained high, reflecting the ever increasing gravity of the underlying causes and associated diseases.
Chronic pyelonephritis
Until well into the 1960s chronic pyelonephritis was a popular diagnosis,11,12 erroneously deemed to be the most common cause of uremia, extensively covered in textbooks and lectures. In retrospect encompassing many different conditions, some secondarily infected in an era when antibiotics were not yet available. Wagner in 1882 had described the so-called pyelonephritic contracted kidney in a report of five cases associated with urethral stricture; students of the subject were referred to the work of Escherich (who in 1894 had described pyelitis of childhood), to Löhlein’s comprehensive paper on contracted kidneys (1917), and to Gibson’s 1928 series of cases with carbuncles, septic infarcts, abscesses, including milder and obstructive cases, mostly with uneven kidneys, patchy lesions interspersed with normal areas, or with infection of the pelvis. In 1930 Stamler and Dopheide had published in Chemnitz, Germany, beautiful illustrations of the pathology of so-called chronic pyelonephritis; and between 1933 and 1937 Longcope described under the same diagnosis cases of bilateral disease; mostly in young white women with recurrent attacks of infection dating from childhood, sometimes with hypertension and usually growing E. coli from the urine. The kidneys were described as uneven, their calyces distorted, the pathology again patchy.
In a further widely quoted publication Weiss and Parker had laid down in 1939 the histological diagnostic criteria for the diagnosis of chronic pyelonephritis, noting that 15–20% of their patients had malignant hypertension. Bell in 1942 reported under the same diagnosis cases with multiple abscesses, renal carbuncles, peripheral abscesses, and ureteral obstruction. In 1953 Kinkaid–Smith made a detailed histological study of cases seen at Hammersmith Hospital in London and concluded that the hypertension of “chronic pyelonephritis” was due to vascular changes. Later Saphir and Diamand claimed that chronic pyelonephritis accounted for most cases of malignant hypertension.4,8
Clinically chronic pyelonephritis was often diagnosed on the basis of irregular renal contours, distorted calyces on retrograde pyelography, interstitial infiltrates on renal histology, or a history of enuresis and pyelitis in childhood, followed by honeymoon cystitis, bladder catheterization during pregnancy, further attacks of acute pyelonephritis, and progression to uremia by the third decade. Pyuria was thought to be characteristic, in some cases elicited by provocative tests with pyrogen or prednisone, and there were well accepted radiologic criteria. Even in the mid-1960s some nephrologists would culture the needle after kidney biopsy, and use antibiotics empirically in the hope of reversing the uremia of chronic pyelonephritis.
Later, with advancing knowledge, the concept of chronic pyelonephritis began to be questioned, notably by Lawrence Freedman in 1967.12 It gradually became apparent that this diagnosis masked a multitude of sins, some patients having reflux nephropathy, others analgesic nephropathy, chronic “benign” hypertensive nephrosclerosis, focal glomerular sclerosis, and various cystic, congenital, and interstitial renal diseases. As a result, the chapter on chronic pyelonephritis has disappeared from modern textbooks of nephrology.
Renal biopsy

Robert M. Kark
Many investigators contributed to the development of this important diagnostic technique. Some of the earliest renal biopsies were surgical for diagnosis of cancer or for decapsulation of the kidney as treatment for glomerulonephritis. Surgical specimens were also available when sympathectomy was used to treat intractable hypertension. Renal tissue was occasionally obtained unintentionally during needle liver biopsy, and there are several conflicting stories on this subject.13–15
The earliest intentional renal biopsies were done in 1944 by Nils Alwall in Lund, Sweden, who biopsied 13 patients but abandoned the procedure after the death of a patient with a single kidney. In 1950 in Cuba, Antonino Perez-Ara attempted renal biopsies and published his results in an obscure and generally unavailable journal. Then in 1951 Claus Brun and Poul Iverson in Copenhagen developed the procedure using an aspiration technique and first demonstrated that renal biopsy had genuine diagnostic utility.13–15
As with most procedures, true priorities are difficult to establish. It suffices to say that other investigators were also active at that time, including Richard Joske (Australia), Alvin Parrish and Richard Howe (USA), Aldo Torsoli and Enrico Fiaschi (Italy), and Maurice Payet (Senegal).14,15 Much of the credit for popularizing renal biopsy must be given to Robert Kark of the University of Illinois at Chicago (1954), who with Robert Muerhcke, Victor Pollak, and Conrad Pirani perfected the technique of renal biopsy in the prone position using the Franklin modification of the Vim-Silverman needle and improved localization of the kidney by intravenous pyelography. They biopsied patients with various diseases, and in classical papers described the morphology of diabetes, lupus nephritis, nephrotic and nephritic syndromes, and toxemia of pregnancy.16–18 They fully described the changes observed in diabetic nephropathy, formulated an early classification of lupus nephritis, and introduced its treatment with high doses of steroids.16–18
The introduction of renal biopsy profoundly increased the understanding of glomerular diseases. Minimal change nephrotic syndrome became recognized as an entity in its own right, as were membranoproliferative (“hypocomplementemic”) glomerulonephritis, focal glomerular sclerosis, and crescentic glomerulonephritis. Thin sections, special stains, immunofluorescent staining, and electron microscopy added precision to diagnosis; and safety was increased with the use of direct fluoroscopy, ultrasound, or computed tomography. Newer disposable needles, including some with automatic gun control, also increased ease and safety, making it even possible to biopsy outpatients.
Dialysis
Thomas Graham (1805–1869) in 1855 used thin parchment to demonstrate diffusion across semipermeable membranes and coined the term dialysis [dia = through, lyein = loose].19–24 In 1913 at Johns Hopkins, Abel, Rowntree, and Turner experimented with the “first extracorporeal device that permitted the diffusion of substances from the blood,” using hirudin from leeches as an anticoagulant and collodion semipermeable membranes to dialyze salicylate-intoxicated dogs. In 1914 Von Hess and McGuigan improved Abel’s apparatus by adding pulsatile flow. In the 1920s G. H. Love experimented with chicken gut membranes as semi-permeable membranes; and Herman Necheles used animal peritoneum membranes to dialyze nephrectomized dogs, again using hirudin as anticoagulant (1923). Further experiments on dogs were carried out by William Thalheimer in New York (1937).19–24

Willem Kolff
In 1923 George Haas of Giessen, Germany, performed the first hemodialysis in man using hirudin and collodion membranes, but abandoned this project because of technical difficulties and “lack of recognition.”25 His work was taken up in 1943 by Willem Kolff, working during the German occupation of the Netherlands in the town of Kampen. Taking advantage of the availability of heparin and cellophane, he built the rotating drum artificial kidney, in which loops of cellophane were wound around a central drum immersed in dialysis fluid. Kolff’s first 29 treated patients all died before he was able to effect a recovery in a comatose patient with acute renal failure. His work was later continued in several other countries, especially in Boston, where his artificial kidney was modified by J. P. Merrill and used in clinical practice as the Kolff-Brigham kidney.26–28
Other dialyzers built in that era include those of Nils Alwal (Lund, Sweden, 1944); Von Garrelts (Stockholm, 1944); George Murray (Toronto, 1947—first dialysis in North America), and Inouye and Engleberg (Philadelphia, 1953—pressure cooker dialyzer). Flat or parallel flow dialyzers were built by McNeil (Buffalo, 1947), Skeggs-Leonard (Cleveland, 1948), and eventually by Frederik Kiil (Oslo, 1960).21 Yet the regular use of dialysis was slow to take off, largely because of its complexity. Then in 1954 Willem Kolff, working at the Cleveland Clinic and acting, as he put it, on the principle that in America everything had to be disposable, wound cellophane tubing around soft drinks cans to build a disposable dialyzer. This was later perfected by Travenol-Baxter as the Twin Coil and used in a steel tank. It had a volume of some 1100 ml, expanded as the internal pressure increased, and needed priming with two units of blood before use.26–28
Dialysis in the early days was a complicated affair resembling major surgery, often with teams of doctors and nurses in attendance, blood stained swabs and used syringes everywhere, as well as frequent and unexpected complications. Some units had an adjacent laboratory for two hourly blood determinations to discover if enough urea had been removed. Access to the vascular system was obtained by cutting down on the forearm vessels, using tapered plastic cannulae that sometimes were heparinized and left in situ until the next dialysis. Cut downs were also needed for the several double lumen catheters developed at that time, such as that of Piazza in Peru. Then in London in 1961 Shaldon adapted the Seldinger percutaneous approach to dialysis by using stiff opaque catheters made in his laboratory. He at first cannulated both the femoral artery and vein, but later modified his approach by inserting two catheters in the same vein. His introduction of regional heparinisation and his first attempt at overnight home dialysis were also significant advances in that early period.29

Chronic or maintenance dialysis became practical with the introduction by Scribner and his colleagues of the external arteriovenous shunt. This was made of different sections of Teflon and silastic; at first held together by metal crimp rings and stabilized on the forearm by a metal plate, but later simplified to single tubes of silastic with Teflon tips.30,31 External shunts were followed in 1967 by the work of Brescia and Cimino, who ingeniously connected a forearm vein with its artery to construct a fistula, thus allowing the veins to become dilated and be readily punctured for access.32,33 The realization that not all patients could be dialyzed through native fistulae led in the 1980s to the development of bovine and synthetic grafts, single and double lumen catheters for subclavian and jugular catheterization, and several prosthetic devices that would obviate the need for repeated venepuncture. Travenol coil dialyzers, widely used in the 1960s, gradually became smaller, their priming volume reduced from an initial 1100 ml to about 250 ml. They were later replaced by the even smaller coil dialyzers of the Extracorporeal Company (about 1967)34 and eventually by hollow fiber dialyzers (1972), at first manufactured from modified cellulose and later from more biocompatible synthetic materials.
In the 1970s dialysis became increasingly more available owing to government-sponsored programs. The last two decades of the century saw the building of large outpatient dialysis facilities, numerous technical advances, the use of dialysis machines with proportioning pumps, improved monitoring devices, bicarbonate dialysis, better antihypertensive drugs, vitamin D derivatives, parenteral iron compounds, and erythropoietin, and more recently attempts to carry out long nocturnal or short daily dialysis. There has also been a growing preoccupation with models predicting adequacy of dialysis, often by complex and not necessarily clinically relevant mathematical models.
Peritoneal dialysiswas first attempted by Ganter in Germany in 1926.35 It became a practical proposition in the late 1950s with the development of commercially available solutions and catheters that could be easily inserted by the bedside, such as the Weston-Roberts catheter. Early attempts to carry out chronic dialysis utilized the implanted Barry button and the removable Dean Jacobs prosthesis, but became more realistic with the development of implantable catheters, of which the Tenkhoff catheter has remained the prototype. Continuous ambulatory peritoneal dialysis and various types of automatic cyclers were the next advance, resulting in a wider application of this technique. The introduction of better systems of dialysis bags and various ways of connecting them to the patients, as well as a better means of treating infection, have resulted in a reduction of the peritonitis rate from 4 to 0.5 episodes per year. There have also been advances in the understanding of peritoneal physiology and of how solutes are transferred across the peritoneal membrane.
Transplantation
The story of renal transplantation begins in Vienna and Berlin, where in the early 1900s several investigators carried out experimental work and transplanted kidneys from one animal species to another.36,37 In 1902 Hungarian-born Emerich Ullman reported the successful autologous transplantation of a dog kidney to the neck. In the same year he performed the first xenotransplantation—a dog kidney to a goat. He also attempted to implant the kidney of a pig into the elbow of a young uremic woman, but encountered technical difficulties and abandoned the attempt.37
Between 1905 and 1910 Ernst Unger in Berlin carried out further experiments on heterotransplantation, at first in animals, but in 1909 he implanted both kidneys of a stillborn human baby into a baboon. The next year he implanted a kidney from a Borneo Macacus monkey into the thigh of a young woman who was deteriorating from renal failure. The kidney produced only a little bloody urine and the patient died soon afterwards. In Lyons, France, Mathieu Jaboulay (1906) also made attempts at heterotransplantation in man, using pigs and goats as donors. Alexis Carrel, working in France and later in New York, perfected a method for anastomosing blood vessels and eventually received the Nobel Prize for his work in vascular surgery.

Dr. John P. Merrill (left) explains the workings of an artificial kidney to
Richard Herrick (middle) and his brother Ronald (right). The Herrick twin
brothers were the subject of the world’s first successful kidney
transplant, Ronald being the donor.
The next attempt at transplantation took place in Paris in 1952, by which time techniques of vascular anastomosis and ureter implantation had improved. On December 18, 1952, Jean Hamburger transplanted a kidney from a mother to her child, Marius Renard—a 15-year-old boy who had fallen from a roof and avulsed the pedicle of a solitary kidney. The graft functioned for 22 days before being rejected.38 The event excited worldwide attention and stimulated other investigators in this field. Two years later John P. Merrill and Joseph Murray in Boston began a program of live donor transplantation in twins. By 1957 they were able to report on seven successful transplants.39
There were also several attempts at transplanting kidneys from unrelated donors, in Boston and in Paris, using total body irradiation or irradiation of the kidney, but the efforts were plagued by rejection. Success came with the introduction of better immunosuppression, first 6-mercaptopurine and corticosteroids, later the use of azathioprine by Calne (1960). Cadaver kidney transplantation, first done at the Hospital Necker in 1963, was soon also carried out by J. P. Merrill and J. Murray in Boston.39 By 1964 cadaver kidneys were also being used by renal units in Richmond, Cleveland, and Los Angeles, soon to be followed by a large transplant program in Denver. With the discovery of the HLA groups and the introduction of tissue typing, renal transplantation became safer and more successful. Further advances were made during the 1960s and 1970s, leading to the modern era of new and effective immunosuppressive agents such as cyclosporin A, tacrolimus, mycophenolate mofetil, sirolimus, and monoclonal antibodies. At present, two-year patient survivals after transplantation exceed 95%, and the mean half life of a renal graft stands at 13–17 years for living donors and 8–10 years for cadaver kidneys. Active research is currently being pursued on many fronts: on xenotransplantation; on developing better drugs, perhaps based on different antibodies, cytokines, or adhesion molecules; and there is also an urgent need to help the millions of people living in developing countries for whom treatment is still not available.40
Conclusion
There can be little doubt that in the past renal disease has caused much illness and suffering. It was mostly not recognized as such because diagnostic methods were not available. Many illustrious persons in history are believed to have had kidney disease.41 Michelangelo suffered from kidney stones and gouty arthritis;42 Samuel Johnson was afflicted by progressive dropsy; and even Richard Bright may have possibly died from the disease he had described. It appears that the Hungarian king Stephan Bathory (1533–1585) and several members of his family had polycystic kidney disease.43 Mozart’s terminal illness, still a subject of controversy, may have been uremia from glomerulonephritis or Henoch Schonlein nephritis.44 A report of the autopsy of Ludwig Van Beethoven (written in Latin) indicates that in addition to liver cirrhosis he also had clear evidence of papillary necrosis, most likely analgesic-induced.45 It has also been suggested that Oliver Cromwell died of pyonephrosis secondary to calculous urinary tract obstruction, with septicemia and renal insufficiency;46 and that Samuel Pepys (1633–1703), author of a diary that has endured to this day, was afflicted by some familial form of nephrolithiasis, possibly urate stones. Then there was Napoleon III, dying in exile in England from uremia consequent to calculous obstructive uropathy. His more illustrious uncle, Napoleon I, also had kidney stones.
In recent years two heads of state, Ferdinand Marcos and Yuri Andropov, developed uremia and had dialysis for some time. At the age of 82 the widely read author James Mitchener entered a dialysis program, at first greatly pleased that he “lucked upon a disease that could be treated.”47 Also remembered in nephrological circles is Clyde Shields, the first long-term survivor treated in Seattle by Belding Scribner and his team. To the many other patients dying prematurely of uremia within living memory, the very thought of dialysis or transplantation would have been inconceivable; and now there is every indication that the future will bring about even more spectacular developments.
References
- Müller, F. Morbus Brightii. Verhandl. d. Deutsch. Path. Gesellsch., 1905;9:64–99.
- Munk, F. Klinische Diagnostik der degenerativen Nierenkrankheiten Ztschr. f. klin. Med. 78: 1–52, 1913.
- Black, Sir Douglas. The story of nephrology. J R Soc Med 1980; 73: 514–518.
- Fogazzi, G. B., Ritz, E. Novel classification of glomerulonephritis in the monograph of Franz Volhard and Theodor Fahr. Nephrol Dial Transpl 1998; 13: 2965–2967.
- Heptinstall, R. Pathology of the Kidney, First edition, Little Brown &Co., Boston 1966.
- Brod, J. The Kidney, London 1973, Butterworths, page 552.
- Oken, D. E. Nosologic considerations in the nomenclature of acute renal failure. Nephron 1971; 8: 505–510.
- Bywaters, E. G. L., and Beall, D. Crush injuries with impairment of renal function BMJ 1:427–432,1941.
- Editorial. Acute Renal Failure. Lancet, 1973,I:134–5.
- Myers, M.D., Bryan, D. and Moran, M.D., S., Mark. Hemodynamically mediated acute renal failure. NEJMed 1986; 314: 97–104.
- Rosenheim, ML. Problems of chronic pyelonephritis. BMJ 1963, i:1433.
- Freedman, LR. Chronic pyelonephritis at autopsy. Ann Int Med 1967; 66: 697–710.
- Iverson, P and Brun, C. Aspiration biopsy of the kidney. Am J Med 1951; 11: 324–330.
- Cameron, JS and Hicks, J. The introduction of renal biopsy into nephrology from 1901 to 1961: A paradigm of the forming of nephrology by technology. Am J Nephrol 1997; 17: 347–358.
- Fogazzi, G. B., and Cameron, J. S. The early introduction of percutaneous renal biopsy in Italy. Kidney Int. 1999; 56: 1951–1961.
- Muehrcke, R. C., Kark, R. M., and Pirani, C. L. Technique of percutaneous renal biopsy in the prone position. J Urol 1955; 74: 267–277.
- Kark, R. M., Pirani, C. L., Pollak, V. E., Muehrcke, R. C., and Blainey, J. D. The nephrotic syndrome in adults: A common disorder with many causes. Ann Int Med 1958; 49: 751–774.
- Muehrcke, R. C., Kark, R. M., Pirani, C. L., Pollak, V. E. Lupus nephritis: a clinical and pathological study based on renal biopsies. Medicine 1957;36:1.
- Schreiner, GE. Dawn of dialysis. ASAIO Journal 1993, 828.
- Fagette, P. Hemodialysis 1912–1945: No medical technology before its time. ASAIO Journal 1999; 45,: 238–249 and 379–391.
- Twardowski, Z. J. History of hemodialyzers’ designs. Hemodialysis International, 208, 12:173-210.
- Gottschalk, C. W. and Fellner, S. K. History of the science of dialysis. Am J Nephrol 1997; 17: 289-298.
- Peitzman, S. J. Origins and early reception of clinical dialysis. Am J Nephrol 1997; 17: 299-303.
- Drukker, W., Parsons, F. M., Maher, J. F. Replacement of renal function by dialysis, Martinus Nijhoff publishers, first edition, 1983.
- Paskalev, D. N. George Hass (1886 to 1971) The forgotten hemodialysis pioneer. Dialysis and Transplant 2001; 30: 829–832.
- Kolff, WJ. First clinical experience with the artificial kidney. Ann Int Med 1965; 62: 608–619.
- Kolff, WJ. The early years of artificial organs at the Cleveland clinic Part I: Artificial kidney and kidney transplantation. ASAIO Journal 1998.
- Dunea, G. Willem Kolff: A great man. Hemodialysis International 2009:13:150–151.
- Shaldon, S. Percutaneous vessel catheterization for hemodialysis. ASAIO Journal 1994.
- Konner, K. History of vascular access for hemodialysis. Nephrology Dial Transplant 2005; 20: 2629–2635.
- Scribner, BH., Buri, R., Caner, J. E. Z., Hegstorm, R., and Burnell, JM. The treatment of chronic uremia by means of intermittent hemodialysis: A preliminary report. Trans ASAIO 1960; 6: 114–122.
- Brescia, M. J., Cimino, J. E., Appel, K., Hurwich, B. J. Chronic hemodialysis using venipuncture and a surgically created arteriovenous fistula. N Engl J Med. 1966 ;275:1089–1092.
- Ronco, C., Blagg, C. R. James E. Cimino M.D.: Recipient of the 2008 International Society for Hemodialysis Belding Scribner Trailblazer Award. Hemodialysis International, 2008:12:S66–S67.
- Bergman, L. A., Basha, N. M., Gara, A. H., Ellison, M. R., Smith, E. C., and Dunea, G. The EX-01 dialyzer cartridge. Experience with 800 dialyses. Trans Amer Soc Artif Intern Organs 1969; 15:65.
- Ganter, G. Ueber die Beseitigung giftiger Stoffe aus dem Blute durch Dialyse. Munch t,1ed Wchnschr 1923; 70: 1478–80.
- Hierholzer, K., and Winau, R. Pioneer nephrologists of Berlin. Am J Nephrol 1992; 12: 442–450.
- Nagy, J. A note on the early history of renal transplantation: Emerich (Imre) Ullmann. Am J Nephrol 1999; 19: 346–349.
- Richet, G. Hamburger’s achievement with early renal transplants. Am J Nephrol 1997;17: 315–17.
- Epstein, M. John P.Merrill: The father of nephrology as a specialty. Clin J Am Soc Nephrol, 2009; 4:2–8.
- Cameron, J. S. The future of nephrology with special regard to advances in treatment. Kidney Int 1992; 42: S-203–214.
- Eknoyan, G. On the renal ailments of creators and leaders. Am J Kidney Dis 1993; 21: 653–658.
- Eknoyan, G. Michelangelo: art, anatomy, and the kidney. Kidney international 2000: 57:1190–1201.
- Torres, V. E., and Watson, M. I. Polycystic kidney disease: from antiquity to the 20th century. Nephrol Dial Transplant 1998, 13:2690–2692.
- Guillery, E. N. Did Mozart die of kidney disease? A review from the bicentennial of his death. J Am Soc Nephrol 1992; 2: 1671–1676.
- Schwartz, A. Beethoven’s renal disease based on his autopsy: the case of papillary necrosis. Am J Kidney Dis 1993;21:643–652.
- Felts, J. H. Pascal’s law and the death of Cromwell. Am J Nephrol 1991; 11: 332–333.
- Rosenblum, A. A unique dialysis quality management experience: An interview with novelist James A Michener. ANNA Journal 1995;22:465.
GEORGE DUNEA, MD, FACP, FRCP, FASN is the president and CEO of the Hektoen Institute of Medicine. He is also a professor of medicine at University of Illinois at Chicago, the medical director of Chicago Dialysis Center, and founding chairman emeritus, Division of Nephrology, Stroger Hospital of Cook County. He also serves as Editor-in-Chief of Hektoen International.
