Jayant Radhakrishnan
Darien, Illinois, United States
It pays to keep an open mind, but not so open that your brains fall out.
– Carl Sagan
Surgical advances typically occur in small, incremental steps. Major changes are resisted vigorously, particularly when children are affected. Occasionally, a seemingly outlandish idea greatly improves first the care of children, and later of adults. The following four concepts are entrenched now, but faced considerable antagonism when they were first introduced.
Cardiovascular surgery
Both Francisco Romero in 1801 and Dominique Jean Larrey in 1810 had drained the pericardium successfully. On July 10, 1893, Dr. Daniel Hale Williams repaired a stab wound to the heart of James Cornish at Provident Hospital, Chicago. The German surgeon Ludwig Rehn repaired a stab wound of the heart on September 7, 1896. Yet the heart and great vessels were considered untouchable until Dr. Robert Gross repeatedly demonstrated that they could be operated upon safely in children.
Gross reported on successful ligation of a patent ductus arteriosus in 1939.1 He was the surgical chief resident when he carried out the operation on a seven-year-old girl on August 26, 1938 while Dr. William Ladd, the chief of surgery at Boston Children’s Hospital, was on vacation. Gross was well prepared. He had repeatedly practiced the operation in the laboratory, but he knew that Ladd would not have let him perform it on a patient. When Ladd learned what Gross had done, he fired him, but then had to hire him back. Ladd never forgot this act of disobedience and spoke against Gross being appointed surgeon-in-chief when he retired. Gross got the job anyway. Years later, after Gross retired, the young patient he’d saved, now fifty-eight years old, visited Gross. He told her, “You know, Lorraine, if you hadn’t made it, I might have ended up here in Vermont as a farmer.”2
Next, in 1945, Gross repaired a coarctation of the aorta. His technique of resection and anastomosis and the use of preserved human aortic homografts for replacement became the technique of choice for aortic aneurysms in adults. That same year, he treated a double aortic arch successfully. This was followed by successful operations on other forms of aortic arch anomalies and aberrant large vessels. He went on to repair many complex cardiac anomalies in neonates, infants, and children. His techniques have all been adapted for use in adults with cardiac anomalies. Before cardiopulmonary bypass was available for children, he would suture a rubber well that he had devised to the atrium. When the atrium was entered, blood from the heart would back into the well and prevent air from being sucked into the heart. Gross operated through the resulting pool of blood, seemingly only by feel.
In 1939, Dr. Helen Taussig approached him with the concept of creating an artificial ductus arteriosus to relieve pulmonary ischemia in tetralogy of Fallot. He disagreed, believing it would flood the lungs; it was left to Dr. Blalock to make the shunt in 1944.3 Gross made a point of letting all his trainees know about this incident and emphasized the need to carefully consider every idea presented to a surgeon, regardless of the source.
In 1941, Ladd and Gross wrote the first textbook of pediatric surgery. It was entitled Abdominal Surgery of Infancy and Childhood. Gross expanded it in 1953 and named it Surgery of Infancy and Childhood, and in 1970 he published an Atlas of Children’s Surgery. He was clearly an exceptionally talented surgeon. His skills were appreciated even more when after retiring he revealed that he had been blind in one eye during his entire surgical career. The following sign hung in his operating room: “If an operation is difficult, you are not doing it properly.”3
Reconstructive urology
In the 1950s, pediatric urology was not a defined specialty and reconstructive urology had not yet been envisioned. While a senior resident at Boston Children’s Hospital with Dr. Gross, Dr. W. Hardy Hendren III saw many patients with various forms of urinary diversions that had been created in the hope that kidney function would improve, but did not. Hendren was instructed by his predecessor that these patients were now his responsibility. When he replied that he did not know anything about urinary tract problems, he was told that neither did anyone else.
When Dr. Gross banished Hendren from Boston Children’s Hospital, he returned to Massachusetts General Hospital and started the pediatric surgical service there. On September 22, 1960, he removed the narrow lower end of an obstructive megaureter and tapered the lower ureter before reimplanting it in the bladder. The operation was successful, and the kidney retained normal function for the next forty-eight years. Later, he tapered a refluxing megaureter. By 1968, he had a series of thirty-three megaureters in twenty-two children that he presented at a meeting of the American Urological Association. A senior urologist questioned his integrity because he could not believe that the pre- and post-operative radiographs belonged to the same patients. However, pediatricians dealing with children with urological problems started referring many patients to him. As he gained experience, he continued to innovate. When he presented a paper entitled “A new approach to the infant with severe obstructive uropathy—early complete reconstruction,” the surgeon who had previously doubted him said, “Anybody who thinks he can do surgery of that magnitude on a small baby must think he can walk on water!” Never at a loss for words, Hendren replied, “I would like to invite him to visit Boston next January when the Charles River is frozen and we’ll walk across it together!”
Next, Hendren successfully “undiverted” numerous diverted urinary tracts. In time, his ideas caught on and led to refunctionalization of the urinary tract and anatomic and extra-anatomic maneuvers to avoid urinary diversions. He found ways to deal with patients with both persistent and exstrophic cloacae without exteriorizing their urinary or gastrointestinal tracts. This was a whole different level of reconstruction.4 The field of reconstructive urology for children and adults evolved from these efforts.5 In 2012, the American College of Surgeons awarded him the prestigious Jacobson Innovation Award.6 On a Friday night that turned into a Saturday morning in the operating room, while Dr. Hendren took down and redid a complicated pyeloplasty for the third time, I learned that one must persist until the final product is perfect. Good enough is not good enough.
Splenic preservation
Until the late 1960s, the spleen was considered dispensable and was removed for the slightest of indications. The adage for all splenic injuries was that it was too friable to repair and prone to delayed ruptures if left in situ. In 1967, the chief of pediatric general surgery at the Hospital for Sick Children, Toronto mentioned to Dr. Purushottam Upadhyaya from the All-India Institute of Medical Sciences, New Delhi and Dr. James Simpson, an attending surgeon at the hospital, that he had recently operated on a six-year-old boy for a bowel obstruction and found the spleen to be lying in two viable halves. The child had a previous history of abdominal trauma; hence, it seemed likely that the spleen fractured transversely down the middle and both halves healed spontaneously. He wondered whether they could research the possibility of spontaneous healing of the spleen.
Upadhyaya and Simpson retrospectively reviewed fifty-two cases of splenic injury. Thirty patients with isolated splenic injury and another twelve with suspected splenic injury survived, while ten with splenic injury associated with other severe injuries died. They learned that the main splenic vessel divided into five to six branches at the splenic hilum with each branch supplying a segment. Lacerations occurred transversely along the vessels and their accompanying trabeculae, but the vessels were usually not injured, and the bleeding stopped spontaneously. They concluded that splenic injuries could be managed nonoperatively, but that was contrary to long-standing surgical dogma.7 Upadhyaya continued to study the subject in the laboratory and demonstrated injury patterns of the spleen.8 Pediatric surgeons got on the splenic salvage bandwagon in about five years,9 but surgeons who dealt with adult trauma victims were vehemently opposed to the approach. Eventually, they too saw the benefits of splenic salvage and it is now attempted in all patients.
Extracorporeal Membrane Oxygenation for respiratory failure
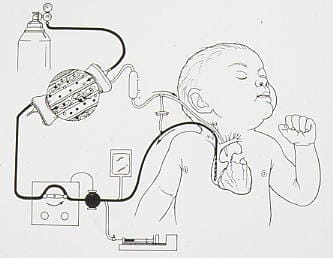
Dr. Robert Bartlett deserves credit for developing and establishing Extracorporeal Life Support (ECLS) as an essential management option in all Intensive Care Units. It has saved lives, initially of neonates and later of older children and adults. Extracorporeal circulation to prolong lives became possible with the development of a silicone semipermeable membrane for exchange of pulmonary gases by Extracorporeal Membrane Oxygenation (ECMO, see illustration). Unlike the previous disc or bubble cardiopulmonary bypass machines, ECMO did not damage erythrocytes, and clotting in the circuit was easier to avoid. It was first tried for Adult Respiratory Distress Syndrome with enough success that the National Institutes of Health funded a prospective multicenter trial. The trial was terminated and it was not revived for years because only 10% of ECMO and control patients survived.
Bartlett first succeeded with ECMO in a two-year-old requiring cardiac support in 1972. In 1975, he successfully treated a neonate with severe respiratory distress due to meconium aspiration. No journal would accept his case report. It was only in 2017 that details of that case were written up.10 Following that one success, Bartlett and his colleagues pressed on and in 1982, they presented a series in which twenty-five of forty-five moribund neonates with respiratory failure survived.11 This encouraged other centers to start using ECMO for neonatal respiratory failure. ECMO was then expanded for older children and later for adults with respiratory failure, provided there was no pulmonary damage. In 1989, the Extracorporeal Life Support Organization (ELSO) was established to keep track of ECMO data internationally. Their data through 2021 demonstrates that when ECMO was used for respiratory problems, 73% of neonates survived, while survival was 61% in older children and 58% for adults. It is worth emphasizing that all these patients were likely to die with conventional treatment.12 COVID-19 patients placed on ECMO had a 39% survival rate.13
Fortunately, none of these pioneering surgeons was subjected to the treatment that Semmelweis received, but they did have to deal with ridicule and disbelief.
References
- Gross RE, Hubbard JP. Surgical ligation of a patent ductus arteriosus. Report of first successful case. JAMA 1939:112;729-731. Reprinted JAMA 1984:251(9):1201-2. doi: 10.1001/jama.251.9.1201.
- Moore FD, Folkman J. “Robert Edward Gross.” National Academy of Sciences. 1995. Biographical Memoirs: Volume 66. Washington, DC: The National Academies Press. doi: 10.17226/4961.
- Hendren WH. Robert E. Gross (1905-1988) and patent ductus arteriosus In ed. Raffensperger JG. Children’s surgery: A worldwide history. 2012 North Carolina, McFarland & Co. Inc. publishers. Chapter 20, pp. 130-139. Bibliography, pp. 291-292.
- Pearson HA. W. Hardy Hendren III MD. 2008. Oral History Project of the American Academy of Pediatrics, Elk Grove Village, Illinois. Interviewed June 16, 2006.
- Radhakrishnan J, Alam S, Kalra P, Gilleran J. Urinary diversion and undiversion. In Bajpai M (ed). Progress in Paediatric Urology, Penwel Publishers PLC, New Delhi, India 2012:14:111-158.
- News from the American College of Surgeons. W Hardy Hendren III receives 2012 Jacobson Innovation Award. Chicago June 8, 2012.
- Upadhyaya P, Simpson JS. Splenic trauma in children. Surg Gynecol Obstet 1968:126:781–790.
- Upadhyaya P, Nayak NC, Moitra S. Experimental study of splenic trauma in monkeys. J Pediatr Surg 1971:6:767–773.
- Upadhyaya P. Conservative management of splenic trauma: history and current trends. Pediatr Surg Internat 2003:19:617-627. 10.1 doi007/s00383-003-0972-y.
- Bartlett RH. Esperanza: The first neonatal ECMO patient. ASAIO Journal. 2017:63(6):832-843. doi 10.1097/MAT.0000000000000697.
- Bartlett RH, Andrews AF, Toomasian JM, Haiduc NJ, Gazzaniga AB. Extracorporeal membrane oxygenation for newborn respiratory failure: forty-five cases. Surgery 1982:92(2):425-433.
- ECLS Registry Report International summary. October 2022. Report data through 2021. Retrieved April 2, 2023.
- Bertini P, Guarracino F, Falcone M, Nardelli P, Landoni G, Nocci M, Paternoster G. ECMO in COVID-19 patients: A systematic review and meta-analysis. J Cardiothoracic & vascular anesthesia. 2022:36:2700-2706.
JAYANT RADHAKRISHNAN, MB, BS, MS (Surg), FACS, FAAP, completed a Pediatric Urology Fellowship at the Massachusetts General Hospital, Boston, following a Surgery Residency and Fellowship in Pediatric Surgery at the Cook County Hospital. He returned to the County Hospital and worked as an attending pediatric surgeon and served as the Chief of Pediatric Urology. Later he worked at the University of Illinois, Chicago, from where he retired as Professor of Surgery & Urology, and the Chief of Pediatric Surgery & Pediatric Urology. He has been an Emeritus Professor of Surgery and Urology at the University of Illinois since 2000.