Jayant Radhakrishnan
Chicago, Illinois, United States
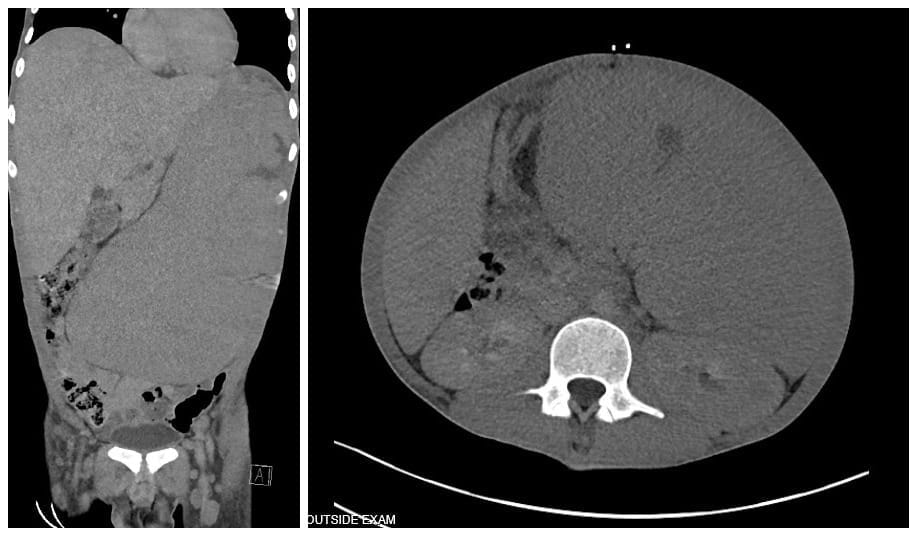
A. The coronal section demonstrates the massive spleen on the left side occupying almost the entire abdomen and the large liver on the right.
B. The transverse section demonstrates the massive spleen.
Patient information and photos courtesy of Dr. Jenna E. Rossoff, Attending Physician (Hematology, Oncology, Neuro-Oncology & Stem Cell Transplantation), Ann & Robert H. Lurie Children’s Hospital and Assistant Professor of Pediatrics (Hematology, Oncology, and Stem Cell Transplantation), Northwestern University Feinberg School of Medicine, Chicago.
Leukemia may have afflicted humans as long as 7,000 years ago,1 but it was not diagnosed until the middle of the nineteenth century. Successful treatment would not be available for another 100 years.
Peter Cullen described “splenitis acutus” with milky blood in 1811, and Alfred Armand Louis Marie Velpeau identified “pus” in the blood vessels in 1825. A report by Jacques Charles Collineau with Gendrin and Caventou, as well as another by A. Duplay, could have been about leukemia. In 1844, Alfred François Donné presented daguerreotypes of microscopic images that demonstrated maturation arrest in leukocytes and differentiated between leukocytes and pus.2 In 1845, John Hughes Bennett described a twenty-eight-year-old patient with massive hepatosplenomegaly and generalized lymphadenopathy who died from suppuration of blood “notwithstanding the absence of any recent inflammation, or collection of pus in the tissues.” He wondered whether the “pus” in the blood was due to hypertrophy of the organs or vice versa.3 He named the condition leucocythemia.4 In a similar case in the same issue of the journal, David Craigie believed the primary problem to be in the blood, the spleen being affected secondarily.3
Rudolf Ludwig Karl Virchow also published a similar case at the same time. A minor dispute about antecedence was settled when Virchow graciously gave credit to Bennett. Virchow believed there was a reversal in the proportion of red to white cells in the blood and there were no pus cells. He called it “white blood,” (German: weisses blut)5 and two years later the more scientific sounding “leukämie” (Greek leukos = white and haima = blood). He distinguished between lymphatic and splenic origins of cells, describing what we now call lymphoma and leukemia respectively.6 Fuller and Cantab were the first to diagnose leukemia in a living person in 1846,7 and in 1868, Ernst Neumann and Giulio Bizzozero independently determined that the problem arose in the bone marrow.8,9
The next advance was the development of a triacid stain by German medical student Paul Ehrlich. It enabled differentiation of granulocytes into eosinophils (acidophils to Ehrlich), basophils, and neutrophils so leukemias could be categorized.10 By 1913, they were classified as chronic myelogenous (CML), chronic lymphatic (CLL), acute myelogenous (AML), and acute lymphocytic (ALL) leukemias.
Southam reported survival times in 150 of 173 children and adults with acute leukemias treated with various modalities at Memorial Hospital, New York, between 1926 and 1948. Half died in seventeen weeks and 90% by thirty-six weeks. The longest survival was fifty-eight weeks.11 Tivey reviewed 428 untreated pediatric acute leukemia patients and found that 50% died within four months of developing the first symptom. Of the rest, 10% lived for eleven months and the middle two-thirds from two to eight months.12
After World War II, nitrogen mustard was used for chemotherapy but it did not work for leukemia.13 The dismal circumstances of children afflicted with ALL, the most common cancer in children, profoundly affected Sidney Farber, a pathologist at Boston Children’s Hospital who had the unenviable task of conducting the autopsies. He decided to switch from studying dead bodies to treating living children despite discouragement by his colleagues, because “it was time that patients, particularly children, with leukaemia were ‘entitled’ to remission.”14 There was some evidence that folic acid inhibited growth of sarcomas and even produced complete regression of breast cancer, so he tried it.15 Farber himself described the unmitigated failure of the experiment as having resulted in an “acceleration phenomenon” of the “leukemic processes to a degree not encountered in his experience with some 200 post-mortem examinations on children with acute leukemia not so treated.”However, he realized “that folic acid antagonists might be of value in the treatment of patients with acute leukemia.” Yellapragada Subbarow supplied him with a pure form of the most potent folic acid antagonist, aminopterin (4-aminopteroic acid), to inject intramuscularly. In ten of the sixteen terminally ill patients, symptoms resolved temporarily along with reduction in the size of the liver and spleen, disappearance of blast cells from the blood, and resumed normal hematopoiesis.16
Farber recognized that to continue this work, public support and capital were essential. He started the Jimmy Fund in 1948 with the Boston Braves of Major League Baseball. When the Braves left for Milwaukee, the Red Sox adopted the charity. “Jimmy” was Einar Gustafson, who had Burkitt’s lymphoma. Farber persuaded Mary Woodard Lasker, the wife of a Chicago advertising magnate; the cardiac surgeon Michael E. DeBakey; Senator J. Lister Hill (Alabama); and Congressman John E. Fogarty (Rhode Island) to help him obtain funds. He more than tripled funding for the National Cancer Institute (NCI) between 1957 and 1967.17 One result of these efforts was development of the Cancer Chemotherapy National Service Center (CCNSC), which experimented with thousands of plant and fermentation products and synthetic chemicals on more than a million mice per year.
Direction was required at NCI, and Charles Gordon Zubrod was hired to provide it. During the Depression, Zubrod dropped out of the College of Holy Cross as he could not afford the tuition. However, the Dean ordered him to return and finish; he received the bill for tuition many years later. Zubrod spent World War II looking for a replacement for quinine, as malaria was the only known treatment for syphilis of the central nervous system at the time. However, persistent fever above 103°F was debilitating, so the malaria was treated intermittently. This work resulted in the development of chloroquine and pamaquine. Working behind the scenes at NCI, he convinced oncologists in various academic institutions to work cooperatively with each other, and he started the NCI Leukemia Service.18 He hired two young researchers, Emil “Tom” Frei III and Emil Freireich, to carry out new and innovative therapies. He also made them use Austin Bradford Hill’s method of randomizing patients in controlled clinical trials instead of continuing the haphazard manner in which studies were reported in the US at the time.19
Frei grew up on the banks of the Mississippi. His mother nicknamed him after Tom Sawyer because too many Emils replied when called. His important concept was that “additional chemotherapy formed a critical component of cure, even when the tumor was in complete remission and no longer detectable.” He also advocated for four-drug therapy with VAMP (vincristine, amethopterin i.e. methotrexate, 6-mercapto purine, and prednisone).20 Freirich, a product of the University of Illinois College of Medicine (UICOM), was fired from the internship at Cook County Hospital for insubordination when he tried to treat a patient relegated to the hospital’s “death room.” He pursued his residency at Presbyterian Hospital until the chief of medicine there was fired for hiring him and the teaching service was abolished. Next, he moved to Massachusetts Memorial Hospital (now Boston Medical Center). When he received a draft notice upon completing his residency, Zubrod offered him a research position at NCI as an alternative to military service, and he took it. His major contribution was the administration of platelets obtained from fresh blood to leukemic children to keep them from bleeding to death. Initially, since the blood bank refused to give blood for his patients, he stole it. Fortunately, Dr. Zubrod supported him and made the bank release the blood.21
Researchers at NCI successfully rid the blood and bone marrow of ALL only for it to relapse in the central nervous system. Donald Paul Pinkel at St. Jude Children’s Research Hospital in Memphis had radical ideas on how to deal with the problem. He named his regimen “Total Therapy”; his trainees knew it as “Total Hell.” He believed that all tumor cells were not sensitive to the same drugs at one time, so he developed multiple drug combinations for a maximal attack and continued chemotherapy for three years. For the nervous system, he instilled drugs into the cerebrospinal fluid (CSF) and also added high-dose radiation to the skull. His sequential clinical studies were put together by incorporating what had been learned in previous studies. In the seventeenth iteration of Total Therapy at St. Jude, they have an ALL survival rate of 94%, and his technique is now also being used in adults.22
Chronic myelogenous leukemia (CML) was also considered a death sentence. In 1882, The Lancet published a letter from A. Cowan [? sic] Doyle from Aston in Birmingham, England about arsenic inducing a remission in a case of leucocythaemia.23 At the turn of the twentieth century, radiotherapy to the spleen improved quality of life without prolonging survival. Internal radiation with radioactive phosphorous followed, and after World War II nitrogen mustard, especially for patients resistant to radiotherapy,13 followed by urethane, busulfan, and hydroxyurea sequentially.24 Allogenic stem cell transplants failed initially, but later, with identical twins and genetically HLA-identical siblings, results improved. Some other techniques used were leukapheresis and autografting.24
A significant advance occurred in 1960 when Nowell and Hungerford studied seven individuals and discovered a small abnormal acrocentric chromosome in chronic granulocytic leukemia cells, while karyotypes of aleukemic cells were normal. This was the Philadelphia (Ph) chromosome, which demonstrated that CML probably originated from a single hematopoietic stem cell.25,26 In 1973, Janet Davison Rowley, a University of Chicago geneticist (who also taught neurology at UICOM), identified it as a mutual translocation between chromosomes 9 and 22.27 Subsequently, she also found a translocation between chromosomes 8 and 21 in AML and between 15 and 17 in acute promyelocytic leukemia.28
Studies determined that unregulated tyrosine kinase activity could activate this BCR-ABL fusion to malfunction like an oncogene. Studies using interferon-ɑ revealed that some patients could become Ph-negative and their life could be prolonged one to two years compared to those treated with hydroxyurea or busulfan. A few patients remained Ph-negative even after therapy was discontinued. In 2001, the development of imatinib mesylate, a tyrosine kinase inhibitor (TKI) that induces complete remissions, was a major step forward.24 Second-line drugs can salvage those that fail with imatinib and once the disease progresses past the chronic stage, allotransplant is an option for eligible patients. The overall five-year survival is 65%, while the five-year relative survival (ratio of observed survival in a population to the expected or background survival rate) is as high as 90%. The final word is still awaited, however, since tyrosine kinase inhibitors do not eradicate CML stem cells.
References
- Scherf H (2015). 7000 years: The oldest case of leukemia discovered. Universität Tübingen, August 31, 2015. https://uni-tuebingen.de/uploads/media/15-08-31_PM_Skelton_Leukemia_EN.pdf
- Kampen KR (2012). The discovery and early understanding of leukemia. Leukemia Research 36(1):6-13. https://doi.org/10.1016/j.leukres.2011.09.028.
- Bennett JH (1845). Case of hypertrophy of the spleen and liver, which death took place from suppuration of the blood. Edin Med and Surg Journal 165.1-1. Edinburgh: Printed by Stark and Company. University of Glasgow Library.
- Bennett JH. Leucocythemia (1845). Clinical lectures on the principles and practice of medicine, 2nd Edition. New York: Samuel S. & William Wood, 1858. p. 79-81.
- Virchow R (1845). I Weisses Blut, Gesammelte Abhandlungen zur wis senschaftlichen medicin. Frankfurt: Meidinger Sohn & comp.; 1856. p. 154-173.
- Virchow R (1856). III. Die Leukämie, Gesammelte Abhandlungen zur wissenschaftlichen medicin. Frankfurt: Meidinger Sohn & comp.; 1856. p. 190-211.
- Fuller HW, Cantab LM (1846). Particulars of a case in which enormous enlargement of the spleen and liver, together with dilatation of all the blood vessels of the body, were found coincident with a peculiarly altered condition of the blood. The London Medical Gazette 23(3):123.
- Spain P, Kadan-Lottick N (2010). Observations of unprecedented remissions following novel treatment for acute leukemia in children in 1948. In: Commentaries on the history of treatment evaluation. James Lind Library. https://www.jameslindlibrary.org/articles/observations-of-unprecedented-remissions-following-novel-treatment-for-acute-leukemia-in-children-in-1948/
- Mazzarello P, Calligaro AL, Calligaro A (2001). Giulio Bizzozero: A pioneer of cell biology. Nature Reviews Molecular Cell Biology 2(10):776-781. doi: 10.1038/35096085.
- Ehrlich P (1909). Beiträge zur experimentellen Pathologie und Chemotherapie. Publisher Leipzig: Akademische Verlagsgesellschaft Collection. Royal College of Physicians of Edinburgh.
- Southam CM, Craver LF, Dargeon HW, Burchenal JH (1951). A study of the natural history of acute leukemia with special reference to the duration of the disease and the occurrence of remissions. Cancer 4(1):39-59. doi: 10.1002/1097-0142(195101)4:1<39:aid-cncr2820040105>3.0.co;2-g.
- Tivey H (1952). Prognosis for survival in the leukemias of childhood; review of the literature and the proposal of a simple method of reporting survival data for these diseases. Pediatrics 10(1):48-59.
- Radhakrishnan J (2025). The history of chemotherapy. Hektoen International Science, Summer 2025. https://hekint.org/2025/07/31/the-history-of-chemotherapy/
- Piller G. (2001). Page 11 in Christie DA, Tansey EM, eds. (2003), Leukaemia, Wellcome Witnesses to Twentieth Century Medicine, vol. 15 (London: Wellcome Trust Centre for the History of Medicine at UCL), Queen Mary University of London. https://qmro.qmul.ac.uk/xmlui/bitstream/handle/123456789/2766/TANSEYLeukaemia2003FINAL.pdf
- Farber S, Cutler EC, Hawkins JW, Harrison JH, Converse Peirce 2nd E, Lenz GG (1947). The action of pteroylglutamicconjugates on man. Science 106(2764):619-621. doi: 10.1126/science.106.2764.619.
- Farber S, Diamond LK, Mercer RD, Sylvester Jr. RF, Wolff JA (1948). Temporary remissions in acute leukemia in children produced by folic acid antagonist, 4-aminopteroyl-glutamic acid (Aminopterin). New Engl J Med 238(23):787-793. doi: 10.1056/NEJM194806032382301.
- Miller DR (2006). A tribute to Sidney Farber-the father of modern chemotherapy. Brit J Haematol 134(1):20-26. doi:10.1111/j.1365-2141.2006.06119.x.
- C Gordon Zubrod, 84, dies; Led chemotherapy research, built cooperative group system. The Cancer Letter 25(4):1-8 January 29, 1999.
- Farewell V, Johnson T (2010). Woods and Russell, Hill and the emergence of medical statistics. Statist Med 29:1459-1476.
- Holland JF, Freireich EJ (2013). Emil Frei III, 1924-2013. J Clin Oncol 31(30):3727-3730. doi. 10.1200/JCO.2013.51.8126; published online ahead of print at www.jco.org September 9, 2013.
- Printz, C (2020). “First person profile: Emil J. Freireich, MD”. Cancer 126 (7): 1373-1374. doi:10.1002/cncr.32812.
- Downing JR, Pui C-H (2022). Obituary: Donald P. Pinkel: In memorium (1926-2022). Blood Cancer Discov 3(4):267-269. doi: 10.1158/2643-3230.BCD-22-0051.
- Cowan Doyle A (1882). Notes on a case of leucocythæmia. Lancet 25th March: 490.
- Goldman JM, Daley GQ (2007). Chronic myeloid leukemia-a brief history. In: Myeloproliferative disorders. Hematologic Malignancies. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-540-34506-0_1.
- Nowell PC, Hungerford DA (1960a) Chromosome studies on normal and leukemic leukocytes. J Natl Cancer Inst 25(1):85-109.
- Nowell PC, Hungerford DA (1960b) A minute chromosome in human granulocytic leukemia. Science 132:1497.
- Rowley JD (1973). Letter: A new consistent chromosome abnormality in chronic myelogenous leukaemia identified by quinacrine fluorescence and Giemsa banding. Nature 243(5405):290-293. doi: 10.1038/243290a0.
- Janet Rowley, cancer genetics pioneer, 1925-2013. UChicago News. Dec. 17, 2013. https://news.uchicago.edu/story/janet-rowley-cancer-genetics-pioneer-1925-2013
JAYANT RADHAKRISHNAN, MBBS, MS (Surg), FACS, FAAP, completed a Pediatric Urology Fellowship at the Massachusetts General Hospital, Boston following a Surgery Residency and Fellowship in Pediatric Surgery at the Cook County Hospital. He returned to the County Hospital and worked as an attending pediatric surgeon and served as the Chief of Pediatric Urology. Later he worked at the University of Illinois, Chicago from where he retired as Professor of Surgery & Urology, and the Chief of Pediatric Surgery & Pediatric Urology. He has been an Emeritus Professor of Surgery and Urology at the University of Illinois since 2000.