Antoinette C. van der Kuyl
The Netherlands
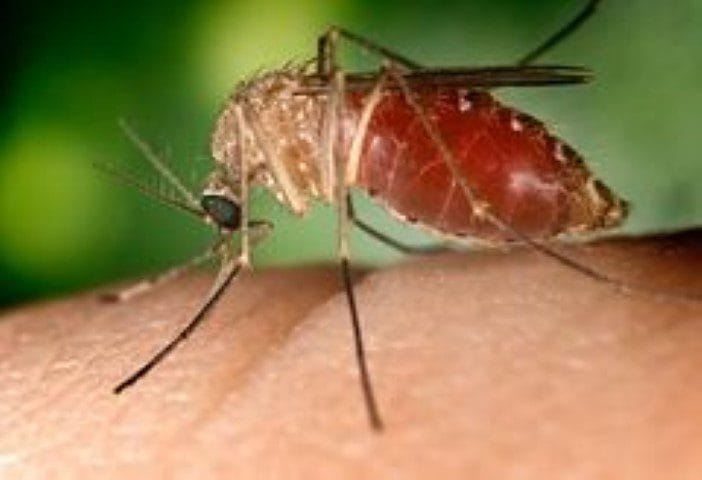
The disease struck New Orleans in July 1828. According to the Boston Commercial Gazette, as reported by the Surinaamsche Courant of September 28, 1828, the streets of New Orleans were full of people using crutches so that the outlook of the city was one of cripples, as the majority of the inhabitants suffered from the novel malady. It had been named Dandy Fever by the people of St. Thomas in the West Indies where almost all 12,000 inhabitants suffered from the affliction in 1827.1 Particularly impressed by the “affectation” displayed by the sufferers, and being a disease unknown to the establishment, they chose this name which translates to “dengue” in Spanish. The name was formally accepted in 1869 by the committee on nomenclature of the Royal College of Physicians and Surgeons of London.2 Dengue reached New Orleans, Louisiana, probably by a vessel from Havana carrying one or two recovering patients.1,3“The weather had been warm, but not exceeding a temperature favorable to health. The river also was of a favorable height, so we were led to expect a healthy season,” according to Philip Dumaresq, who wrote an account of the outbreak that involved himself.3,4 However, this was not to be, as the disease appeared so suddenly, and spread with such rapidity, that at first it caused universal alarm, and was considered a sort of plague that would probably ravage the whole country.1 Fortunately, it was soon discovered, that although a very painful, it was by no means a dangerous disease.1 Indeed, dengue fever (DF) is usually not lethal, but in a minority of cases it can progress to dengue haemorrhagic fever (DHF) and, more seriously, to dengue shock syndrome (DSS), both of which can be fatal.5
During the 19th century DF travelled together with its human hosts and spread throughout the tropical and subtropical areas of the world, first causing outbreaks aboard ships and in coastal towns, and then using railways to reach more remote areas.3 Gradually, the disease also increasingly displayed its more severe manifestations.5,6 Possibly, DHF and DSS can be attributed to antibody-mediated enhancement (ADE) of infection, which means that antibodies that are present from an earlier infection enhance the symptoms of a next infection.7 ADE, as well as limited cross-protection between serotypes, still precludes the development of an effective vaccine against DF.8
To find out more about the aetiology and transmission of DF, the US Army Medical Corps sent two young officers, Captain Ashburn and Lieutenant Craig to the Philippines where an outbreak of DF occurred in 1906. They showed that DF is caused by a filterable agent,9 a microorganism that will pass through filters that have pores too small to allow “ordinary“ bacteria through.10 Unfortunately, they used Culex fatigans (the southern house mosquito, now Culex quinquefasciatus) mosquitoes to test their transmission hypothesis but failed as this species is refractory to infection with dengue. After the 1916 DF outbreak in Murwillumbah, New South Wales, Australia, it was realized that the illness was associated with the abundance of two species of mosquitoes in town, not only Culex fatigans but also Stegomyia fasciata (now renamed Aedes aegypti).11 Experimental work then showed that the latter species, the yellow fever mosquito, could indeed transmit the disease to human volunteers,11 as could other Aedes species. Aedes albopictus is currently adapting to temperate zones suggesting that the virus will soon extend its range.12 In 1952, dengue fever virus (DENV) particles with a rod-like form characteristic of flaviviruses, were first detected by electron microscopy in infected mouse brain.13 Serology combined with viral genome analysis showed that four serotypes of DENV exist, and that the cross-species transmission from monkeys to humans occurred recently between 125 (genotype 1) and 320 (genotype 2) years ago.5
DENV is just one of the viruses that seized the opportunity offered by a vastly expanding human population and its faster ways of transport to conquer the world. About a century ago, the immune-system destroying AIDS virus also left the tropical forests of Africa and its primates to invade a new species and start a pandemic. Although the viruses are from different families, transmit differently (biting insects versus sexual transmission), and differ in virulence, they both play intricate tricks on our immune system. As a result, no effective vaccine has been developed yet despite intense efforts.
Note
Based on the long-lasting arthralgia described, it has been speculated that the outbreaks of DF in the West Indies and Southern States in 1827-1828, as well as the DF reported from India during 1824-1825 were actually cases of Chikungunya, a disease caused by a related virus that is also transmitted by Aedes mosquitos.6,14,15
Editorial note
As of September 2023, at least one dengue fever vaccine is currently in use. See Stephen Thomas, “Is new dengue vaccine efficacy data a relief or cause for concern?” NPJ Vaccines 8, no. 55 (2023), https://doi.org/10.1038/s41541-023-00658-2.
References
- George W. Stedman, “Some Account of an Anomalous Disease which raged in the Islands of St. Thomas and Santa Cruz, in the West Indies, during the Months of September, October, November, December, and January, 1827-8.” Boston Medical and Surgical Journal 1 (1828):662-669. This journal is now The New England Journal of Medicine.
- J.G. Thomas. “Dengue.” Public Health Papers and Reports 6 (1880):136-153.
- Philip J. Dumaresq, “An Account of Dengue, Danga, or Dandy Fever, as It Occurred in New-Orleans,” Medical and Surgical Journal 1 (1828):497-502. DOI: 10.1056/NEJM182809230013201.
- “Papers on the dengue.” The Western Journal of the Medical and Physical Sciences 2 (1828):428-442. November issue, contains papers on Dengue in Charleston by S. Henry Dickson; the History of Dengue in New Orleans by Philip J. Dumaresq, and Dengue Fever in the West Indies by Richard Tuite. There are also “Notices concerning the Dengue or Spanish Fever” on pages 363-367 of this edition. Retrieved from books.google.com.
- Edward C. Holmes, S. Susanna Twiddy, “The origin, emergence and evolutionary genetics of dengue virus. Review.” Infection, Genetics and Evolution 3 (2003):19–28.
- Goro Kuno, “Emergence of the Severe Syndrome and Mortality Associated with Dengue and Dengue-Like Illness: Historical Records (1890 to 1950) and Their Compatibility with Current Hypotheses on the Shift of Disease Manifestation.” Clinical Microbiology Reviews 22 (2009):186–201. doi:10.1128/CMR.00052-08.
- Maria G. Guzman, Mayling Alvarez, “Secondary infection as a risk factor for dengue hemorrhagic fever/dengue shock syndrome: an historical perspective and role of antibody-dependent enhancement of infection.” Archives of Virology 158 (2013):1445–1459. DOI 10.1007/s00705-013-1645-3.
- Thomas SJ, Endy TP. “Current issues in dengue vaccination.” Current Opinion in Infectious Diseases (2013) 26:429-34. doi: 10.1097/01.qco.0000433310.28771.cc.
- Ashburn PM, Craig CF, “Experimental investigations regarding the etiology of dengue fever.” Journal of Infectious Diseases 4 (1907):440–75.
- S. B. Wolbach, “The filterable viruses, a summary.” The Journal of Medical Research 27 (1912);1-25.
- J. Burton Cleland, Burton Bradley, W. McDonald, “Dengue fever in Australia. Its history and clinical course, its experimental transmission by Stegomyia fasciata, and the results of inoculation and other experiments.” Journal of Hygiene 14 (1918):317-418.
- Hanson SM, Craig GB Jr.”Cold acclimation, diapause, and geographic origin affect cold hardiness in eggs of Aedes albopictus (Diptera: Culicidae).”Journal of Medical Entomology 31(1994):192-201.
- Reginald L. Reagan, A.L. Brueckner, “Studies of dengue fever virus by electron microscopy.”” Journal of Bacteriology 64 (1952):233-235.
- D.E. Carey, “Chikungunya and dengue: a case of mistaken identity?” J. Hist. Med. Allied Sci. 26 (1971):243–26.
- Olivia Brathwaite Dick, José L. San Martín, Romeo H. Montoya, Jorge del Diego, Betzana Zambrano, Gustavo H. Dayan, “Review: The History of Dengue Outbreaks in the Americas.” American Journal of Tropical Medicine and Hygiene 87(2012):584–593.
- DOI:10.4269/ajtmh.2012.11-0770.
ANTOINETTE C. VAN DER KUYL, PhD, obtained a master degree in Chemistry at the Free University of Amsterdam and a PhD in Biochemistry at the University of Leiden, the Netherlands. She left the subject of plant virology to study human (and other primate) viruses at the University of Amsterdam (Amsterdam, the Netherlands), where she joined the Department of Medical Microbiology of the Academic Medical Center, first as a postdoc, then as an assistant professor. She has published more than 50 research papers, and teaches courses in Medical Microbiology and Infectious Diseases at the University of Amsterdam and the Amsterdam University College to graduate and undergraduate students.
Highlighted in Frontispiece Volume 7, Issue 3 – Summer 2015
