David M Morens; Gregory K Folkers, and Anthony S Fauci
Bethesda, MD, USA
This article was first published in Lancet Infectious Diseases, Volume 8, Issue 11, Nov. 2008, pages 710-719
Summary
Emerging and re-emerging infectious diseases, and their determinants, have recently attracted substantial scientific and popular attention. HIV/AIDS, severe acute respiratory syndrome, H5N1 avian influenza, and many other emerging diseases have either proved fatal or caused international alarm. Common and interactive co-determinants of disease emergence, including population growth, travel, and environmental disruption, have been increasingly documented and studied. Are emerging infections a new phenomenon related to modern life, or do more basic determinants, transcending time, place, and human progress, govern disease generation? By examining a number of historically notable epidemics, we suggest that emerging diseases, similar in their novelty, impact, and elicitation of control responses, have occurred throughout recorded history. Fundamental determinants, typically acting in concert, seem to underlie their emergence, and infections such as these are likely to continue to remain challenges to human survival.
| Panel.
Factors involved in infectious disease emergence[4], [5] and [6] Often differing for newly emerging, re-emerging, and deliberately emerging diseases, these selected factors include genetic, biological, social, political, and economic determinants
|
Introduction
Not a single year passes without [which]…we can tell the world: here is a new disease!
Rudolf Virchow, 1867 1
Infectious diseases are responsible for 15 million (26%) of 57 million annual deaths in a global population of 6.2 billion,2 a proportion that could rise substantially as chronic diseases continue to be reclassified as infectious—eg, cervical cancer (human papillomavirus), Kaposi’s sarcoma (human herpesvirus 8), and Helicobacter pylori ulcers, among others.
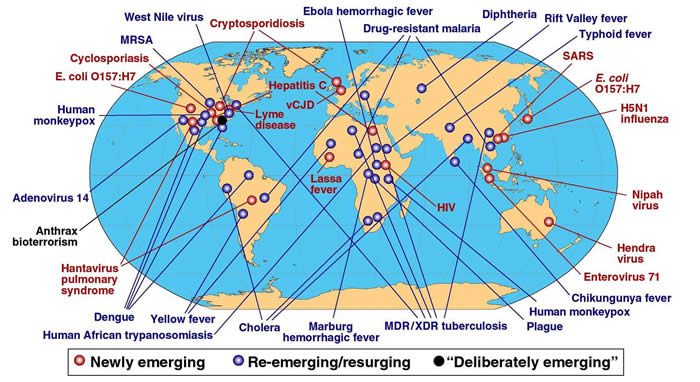
In recent years, the terms “emerging” (ie, newly recognised) and “re-emerging” (previously recognised) infectious diseases have entered the vocabulary of medical science.[3], [4] and [5] These infections also include “deliberately emerging diseases”—eg, bioterrorism.5 Concern about emerging infections has grown following the appearance of new diseases, such as HIV/AIDS, and the re-emergence of others, such as dengue, and from appreciation of the complex determinants of their emergence—eg, microbial adaptation to new hosts (HIV infection, severe acute respiratory syndrome [SARS]), population immunity pressures (influenza A), travel (acute haemorrhagic conjunctivitis), animal migration and movement (West Nile virus infection, H5N1 avian influenza), microbial escape from antibiotic pressures (multidrug-resistant and extensively drug-resistant tuberculosis), mechanical dispersal (Legionnaires’ disease), and others (panel, figure 1).[3], [4], [5], [6] and [7]
Figure 1. Newly emerging, re-emerging/resurging, and deliberately emerging diseases
Selected emerging diseases of public-health importance in the past 30 years (1977–2007), [2] and [5] with representative examples of where epidemics occurred.
Clearly, however, the terminology of emergence is not entirely new.[8] and [9] In the Old Testament, ancient China, classical Greece and Rome, and in subsequent centuries, new diseases have repeatedly been said to be “introduced”, or to “appear”, “approach”, “arise”, “breed”, “progress”, “spread”, or “invade” etc.[10], [11] and [12] In this article we ask two basic questions. Are emerging infections a new phenomenon? And do fundamental determinants, transcending time, place, and human progress govern their generation?
Here we compare several historically notable emerging infections—selected non-systematically to include examples deemed representative of a range and complexity of circumstances, stretching over more than two millennia—to modern emerging infectious diseases and consider the circumstances of their emergence. Since these circumstances seem to have changed little over centuries (see panel, table, and figure 2),[13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26] and [27] it would suggest that fundamental determinants may underlie the seemingly arbitrary emergence of new infectious diseases.
Table. Examples of epidemic emerging infections of historical interest
| Emerging disease | Causal agent | Estimated number of human deaths | Disease factors (see panel) | |
|---|---|---|---|---|
| 430-426 BCE | Plague of Athens13 | Unidentified | 40,000 | 2,5,7,9,11 |
| 1340s | Black Death 14 | Yersinia pestis | 50 million | 2, 5, 6, 7, 8, 10, 11, 13 |
| 1494-99 | French pox (syphilis) 15 | Treponema pallidum | >50 000 | 1, 2, 5, 7, 11 |
| 1520-21 | Hueyzahuatl (smallpox) 16 | Variola major | 3.5 million | 2, 7, 10, 11, 13 |
| 1700s | European cattle epizootics 17 | Rinderpest virus, foot and mouth disease virus, Bacillus anthracis | >15 000* | 4, 5, 6, 7, 8, 9, 10, 11, 13 |
| 1793-98 | The American plague 18 | Yellow fever virus | 25 000 | 2, 3, 4, 5, 6, 7, 8, 9, 10, 12 |
| 1832 | 2nd cholera pandemic, Paris, 19 | Vibrio cholerae | 18 402 | 3, 5, 7, 8, 10 |
| 1875 | Fiji virgin soil epidemic20 | Measles virus | 40 000 | 2, 3, 5, 7, 9, 10, 11, 12 |
| 1918-19 | Spanish influenza 21 | H1N1 influenza virus | ≥50 million | 1, 2, 5, 7, 11 |
| From 1981 | AIDS pandemic 22 | HIV | >25 million | 1, 2, 4, 5, 7, 8, 9, 10, 12 |
Mortality estimates are generally approximations based on speculative data. The numbers in the disease factors column refer to factors associated with disease emergence, as listed in the panel, and apply only to the specific epidemics discussed here. Human disease caused by rinderpest is not known, and by foot and mouth disease virus is uncommon and rarely serious; human disease associated with anthrax was continually observed during the epizootics of the 18th century; one epidemic alone (in Saint-Domingue, Hispaniola) is said to have caused 15 000 deaths.23
The Plague of Athens, 430–426 BC
The [pestilential disease] originated, so they say, in Ethiopia in upper Egypt, and spread from there into Egypt itself and Libya and much of the territory of the King of Persia. In the city of Athens it appeared suddenly…As to the question of how it could first have come about or what causes can be found…I must leave that to be considered by other[s]…
Thucydides, on the Plague of Athens13
The above passage is taken from one of the most famous accounts of an emerging infection ever written: Thucydides’ chronicle of the Plague of Athens, which heralded the end of the Golden Age of Greece (figure 2).[13] and [28] The aetiology of this fatal, complex, multisystemic disease has not been conclusively identified; hypotheses include anthrax, smallpox, typhus, and more than 20 other infectious candidates.28 Thucydides’ vivid account nevertheless represents the first comprehensive clinical-epidemiological characterisation of an infectious disease and the first unambiguous description of an emerging infection. Noting universal susceptibility, multiple independent importations, and an association with war, crowding, and the breakdown of public-health measures, Thucydides also distinguished between signs, symptoms, complications, and variant clinical courses, and presented attack and case-fatality rates. His compelling clinical, public-health, and societally contextual description of an emerging disease was studied by virtually all western and middle eastern medical students for centuries, and profoundly influenced concepts of, approaches to, and even terminology of emerging infections well into the 19th century.
The Black Death, 1347–50
I think it may be said of the plague, as is said of the wind, that it bloweth where it listeth, and wee heare the sound thereof, but know not where it cometh nor where it goeth…
William Boghurst, circa 1665 29
The Black Death of the 14th century (bubonic/pneumonic plague, from the Latin plaga, a blow or stroke) killed approximately 34 million Europeans and 16 million Asians, placing it among the most fatal emerging infections in recorded history.30 The disease’s origin was traced to China in the 1330s or even earlier, from where it proceeded inexorably westward to Europe along caravan and shipping routes.[14] and [30] The many inter-related determinants of, and risk factors for its emergence (table) have been exhaustively documented and discussed (for example, in reference 26).
Accounts of mass deaths, deserted towns, rotting corpses, and free-roaming farm animals are documented in countless texts, and survive today in many works of art (figure 2). The plague is said to have spawned the Tanzwuth (dancing mania) epidemics of the next two centuries, in which bands of itinerant youths roamed the countryside drinking, dancing frenetically, and tossing each other into the air.[31] and [32]
Figure 2. Examples of modern and historically important emerging infectious diseases
-
- (A) Emerging epidemics caused by war and famine. Plague in an Ancient City, by Michael Sweerts, circa 1652, represents the Plague of Athens.13 Oil on canvas, Los Angeles County Museum of Art, CA, USA. The infamous epidemic, the cause of which is still unidentified, occurred during the Peloponnesian wars between Athens and Sparta (430–426 BC).
-
- (B) Emerging epidemics associated with intent to harm. The Black Death (bubonic/pneumonic plague) of 14th century Europe was associated with a bioterrorist attack at Caffa.[24], [25] and [26] The untitled and anonymous painting has been referred to as Death Strangling a Victim of the Plague (circa 1376; Clementinum Collection of Tracts by Thomas of Stitny, also known as Stitny Codex, University Library, Prague, Czech Republic). Photo credit Werner Forman, Art Resource/NY.
-
- (C) Emerging epidemics due to travel and trade. Honoré Daumier’s depiction (hand-tinted woodcut) of the 1832 Paris choléra-morbus epidemic,27 which spread slowly from Asia to Europe along established travel routes.19 National Library of Medicine, History of Medicine Division.
-
- (D) Emerging epidemics associated with microbial adaptation and change. A Maori (New Zealand Polynesian) cenotaph (monument honouring the dead) at a marae (meeting place), Te Koura, New Zealand, memorialises those who died in the 1918–19 influenza pandemic. Photographed by Albert Percy Godber. Cenotaph designed and carved by Tene Waiter. Alexander Turnbull Library, National Library of New Zealand, Wellington, New Zealand.
Multiple independent importations of the plague prompted public-health action in Europe and neighbouring areas. Frightened citizens began isolating arriving ships for as long as 40 (quaranta) days, the origin of quarantine.14 In a Europe emerging from feudalism, government was now held accountable for public health. “[E]veryone must reflect on and prevent the causes that can produce a universal or local pestilence [an epidemic or an outbreak]”, wrote Jacme d’Agramont in 1348. “And if perchance these can be removed they must be removed. And to this end an effort should be made by the Lords [municipal officials] and their officers whose duty it is to look after the…well-being of the community”.33
The Black Death may also have been associated with one of the earliest recorded examples of bioterrorism.[24], [25] and [26] In 1346 a Tartar army besieging the garrisoned Genovese city of Caffa (now Feodosiya, Ukraine) was devastated by plague. The Tartars reacted by catapulting “mountains” (probably thousands) of plague corpses into the city,[24] and [25] allegedly transmitting it to the besieged Genovese.26 600 years later, during World War II, Japanese troops were less successful in starting a plague epidemic by dropping paper bags filled with infected fleas on Chinese cities.[34] and [35]
When (Old and New) Worlds collide: the French pox, 1494
…nowadays we see maladies unknown to our forefathers springing up around us.Ullrich von Hutten, 1519 36
From the 15th through to the 19th century, a time when previously isolated continents were discovering each other and thereby exchanging microorganisms, re-emergence of epidemic diseases associated with geographic spread of microbes became common. In December, 1494, a new disease emerged in Italy;[15], [36] and [37] prostitutes from Naples soon infected soldiers from an invading French army. By July the disease was being diagnosed in mercenaries from Flanders, Gascony (southwest France), Switzerland, Italy, Spain, and elsewhere; they eventually returned home and spread it throughout Europe.
The French pox (syphilis) shocked and terrified.36 “What other contagion has ever spread so quickly to all the countries of Europe, Asia, and Africa?” asked Desiderius Erasmus (“of Rotterdam”); “What other contagion takes such a hold of the entire body…and tortures so cruelly…combin[ing] all that is dreadful in other contagions?”38
Syphilis greatly influenced Fracastoro’s theory of contagion,[39] and [40] the cornerstone of contagionism for the next 300 years. The epidemic also prompted substantial lay thought about disease emergence. Popular explanations (God’s wrath, well poisoning, copulation with monkeys) seemed inadequate for a disease that was obviously acquired by intimate human contact and spread by human movement. In a nosological “epidemic” pre-dating the “Spanish flu” by four centuries, Portuguese, Dutch, and north Africans called syphilis “the Castilian” or “the Spanish” sickness. To Muscovites it became “the Polish sickness”; to the Poles “the German sickness”; to the Germans “the French sickness”; to the French “the Neopolitan sickness”, and so on. Several years passed before a connection was made between the appearance of the French pox and the return of Christopher Columbus from the New World (March 31, 1493), but strong evidence for New World exportation was eventually provided by testimony from Columbus’ men41 and by physicians’ accounts that Hispaniola natives had been affected by the disease since ancient times.42
Societal responses to syphilis in the 15th and 16th centuries were not unlike responses to AIDS 500 years later.43 Undeniable sexual transmission made syphilis one of the few diseases of that era, including leprosy, for which contact contagion was certain. Priests denounced the wickedness and immorality of an age that had provoked God’s anger in the form of a deadly pandemic. Affected spouses, lovers, family, and friends were shunned and abandoned. The hospital bed linens of Parisian patients were initially burned. Soon, victims were turned away entirely; then they were given 24 hours to isolate themselves or leave the city on pain of death by hanging. Charlatans with quack remedies, often painful and deadly, appeared everywhere to treat desperate and dying victims. Nothing worked. Syphilis and humanity therefore had to accommodate each other, and are still doing so 500 years later.
Microbial exchange and disease emergence in the age of discovery: Hueyzahuatl (smallpox), 1520
After our fathers and grandfathers succumbed…dogs and vultures devoured the bodies. So it was that we became orphans, oh, my sons! So we became when we were young. All of us were thus. We were born to die!
Cakchiquel Mayan on the hueyzahuatl epidemic of 1520–21 44
The 1493 European importation of syphilis from the New World did not imply unidirectionality of biological conquests. Spanish adventurers under Hernando Cortés landed on the Yucatan Coast 25 years later and marched to the capital of Tenochtitlan (now Mexico City), humiliating and imprisoning the Aztec ruler Moctezuma. Before Cortés could defeat rival Spanish soldiers sailing from Cuba to restore Moctezuma’s authority, the rival soldiers landed a former slave infected with smallpox. Over the next year the hueyzahautal (great eruption) raged over central America.[44], [45], [46] and [47]
Although smallpox may have first emerged in central Africa 5000 years previously—when an animal orthopoxvirus jumped into human beings48—the disease had probably not reached the New World. Historians believe that about 3.5 million people in central Mexico died in the first year of the hueyzahautal. By the end of the century some 18•5 (74%) of the 25 million population had died,49 presumably largely because of smallpox and additional imported diseases. Smallpox spread southward into South America, ultimately destroying two great civilizations, the Aztec and Inca empires, facilitating Spanish conquests that greatly altered history. Francisco Pizarro, who continued Spanish conquests of South America in the 1530s, is alleged to have undertaken a bioterrorist attack on native peoples using smallpox-contaminated blankets.50 Mexico became a regional geographic reservoir for smallpox and was the source of repeated exportations until the 1940s.
Lessons from emerging epizootics, 1709–99
If there is, within medicine, any subject worthy of…investigation…it is, without doubt, epidemic pestilential diseases, obscure and hidden in their causes, rapid in their progression, frightening in their symptoms, and deadly in their consequences.
Félix Vicq-d’Azyr, 1776 51
The emergence of devastating cattle epizootics in 18th century Europe had a profound impact on concepts of, and responses to, human disease emergence.17 European epizootics of rinderpest,51 a highly contagious bovine paramyxovirus disease, appeared in 1709, to be followed soon thereafter by epizootics of anthrax (1712) and foot and mouth disease (1755).[17] and [52] Between 1709 and 1769, more than 200 million western European cattle died of rinderpest alone, equivalent to 20% of all dairy cows. Rinderpest elicited relatively sophisticated germ theories17 such that immunisation against it was considered 8 years before the first western immunisation was introduced in 1720 (smallpox inoculation).[52] and [53] Notably, rinderpest disease was understood clinically and epidemiologically by about 1770, more than a century before the causal agent was identified and characterised.17
The epizootics of rinderpest, anthrax, and foot and mouth disease led to comparative animal study of disease transmission, to the establishment of the world’s first veterinary schools, and to the first international human disease surveillance/outbreak investigation system.17 Directed by the prominent epizootic investigator Félix Vicq-d’Azyr, surveillance for emerging diseases relied upon field reports of diseases and their suspected risk factors.[51] and [54] When outbreaks occurred, physicians or veterinary students were dispatched to investigate and implement control measures.[17] and [54]
Anthrax has long been associated with intent to harm. More than two centuries before the 2001 anthrax attacks in the USA,55 unscrupulous farmers in Saint-Domingue, Hispaniola, intentionally sent to market the meats of infected livestock, producing in 1770 the deadliest anthrax epidemic on record. 15 000 people are said to have died of the disease, and another 15 000 from the slave revolt it precipitated.[23] and [56] During World War I, a German agent set up a laboratory in Chevy Chase, MD, USA (a mile from the current National Institutes of Health, and several miles from the White House), to make anthrax and other microbial weapons that were then used by paid agents to infect US and Romanian cavalry horses.35 In World War II, Japanese scientists undertook a human bioterrorist campaign with anthrax and other agents.35 A Sverdlovsk (Yekaterinburg, Russia) bioweapon facility accidentally released anthrax spores in 1979, killing more than 60 people.57
The American plague (yellow fever), 1793–98
….much light might be thrown on the causes of violent epidemics, by a series of careful observations, on their beginning and progress…about the time the yellow fever made its appearance in this city…it was observed that musquetoes invaded us in numberless swarms…
Noah Webster, Dec 1 and 6, 1797 10
6 months after delivering his 1793 inaugural address, US President George Washington was fleeing a deadly emerging epidemic. The entire federal government, along with physicians, merchants, and the middle and upper classes, quickly followed. Philadelphia, then the nation’s capital, became a ghost town with an empty harbour, deserted streets, shuttered shops, and rotting corpses.18 Within a few weeks yellow fever, or as Europeans called it “the American plague”, killed more than 5000 of about 50 000 Philadelphians.58 The disease then moved to the major US cities, including Baltimore, Boston, and New York, and to many smaller towns.[18] and [59] Recurrent yellow fever epidemics panicked the nation for six consecutive years, affecting nearly every American in some way.[18], [60] and [61]
Philadelphians who remained in the city formed an emergency government, mobilising the most marginalised citizens—immigrants and African-Americans—mistakenly believing the latter to be naturally resistant.[18], [62] and [63] Opposition by physicians to the use of quarantine measures put them in the middle of angry political debates.[18] and [58] National days of thanksgiving and prayer expressed the general belief that the epidemics were “a national judgment, and the[ir] removal…a national blessing”.64
In 1793, a young belle, Dolley Todd, watched both her husband and infant son die in her arms of yellow fever, then nearly died of it herself. 20 years later the remarried Dolley Madison nursed her second husband—President James Madison—when he nearly died in office of what might have been yellow fever.60 Historians have claimed that deep social and political fault lines were opened up by the American plague of 1793–98, stamping the nation’s character and “[shaking] the newly laid foundations of the Republic…[so that] the crises they aroused have never been entirely put to rest”.58
But the epidemics also led to important advances in public health, such as the establishment by Congress in 1798 of the Marine Hospital System,65 the forerunner of the US Public Health Service. In 1797, America’s first medical journal, The Medical Repository,66 was founded by three physicians who had survived the epidemic. By emphasising emerging epidemics and in publishing bills of mortality and other epidemiological information, the new journal foreshadowed by two centuries the modern journal Emerging Infectious Diseases. In 1799, future lexicographer Noah Webster expanded his yellow fever research into a two-volume text on emerging pestilential diseases,[10] and [67] which influenced epidemiological thought in Europe and the USA for nearly a century.68
“Modern” epidemiology joins emerging disease prevention: cholera, 1832
France and its capital have been visited by a fearful pestilence…It comes without any known cause; it disappears without any revealed reason. The bodies of its victims are in vain examined; death is interrogated: death betrays nothing.
Louis-François Benoiston de Châteauneuf, 183469
In 1830 cholera began to creep slowly from Asia towards Europe along major waterways. It reached Archangel, Russia, in May, 1831; St Petersburg in June; Constantinople at the beginning of July; Pesth (Austro-Hungarian Empire) in mid-July; and Vienna and Berlin at the beginning of August.
The French mounted an unprecedented response.[19] and [70] On March 4, 1831, an interdisciplinary commission of the Académie royale de médecine was appointed; teams of scientists were sent to Russia and Poland to observe cholera first-hand. Coastal health agencies and new quarantine stations were set up; 35 health inspection offices and commissions in the départements (administrative divisions) were established as well.
Paris set up a 43-member central health commission on Aug 20, 1831, 12 subordinate commissions in the 12 arrondissements (districts), and 48 commissions in the quartiers (quarters). The commissions were charged to (1) surveil, investigate, and report; (2) check private and public buildings for sewage connections, wells, cesspools, latrines, and outhouses; (3) construct fountains, gutters, and public latrines; (4) educate the public; and (5) provide charitable services for the poor. Teams of constable-observers helped physicians undertake house to house “sanitary surveillance”.
Despite all of these actions, on March 29, 1832, choléra-morbus exploded violently in Paris (figure 2).[19] and [70] Within a week, incident cases had reached 500 a day, and over 1000 after 2 weeks, 85% of which were fatal. So many deaths were occurring that victims could neither be certified by physicians, nor transported, nor buried fast enough. The government was forced to abandon prevention measures and shift into crisis management.
Thousands of bodies were placed three layers deep in mass graves, which sent a frightening stench over large parts of the city. Corpse wagons were so hastily and shoddily built that they spilled corpses, viscera, and putrid fluids into the streets.19 When furniture vans had to be requisitioned to remove thousands of bodies, citizens were horrified to see layers of corpses piled upon them, claiming they heard cries and sobs coming from still-living victims. Rumours of mass poisoning of the water supply led vigilante mobs to roam the streets searching for suspected poisoners, attacking and sometimes killing innocent people.
Case lists published in the daily newspapers allowed “armchair epidemiologists” to follow mortality and morbidity trends. For the first time in history, a large-scale emerging epidemic was scientifically investigated in “real time”, using census data in a prospective population-based approach that featured analyses of morbidity and mortality stratified by age-group, sex, occupation, socioeconomic status, and location, as well as calculation of case-fatality ratios, age-specific incidence rates, and attributable risks.19 “Modern” epidemiology was born during the 1832 epidemic, but was unable to stop cholera emergence.
The virgin soil epidemic (measles), Fiji, 1875
…The spirits flow away like running-water, au nanuma. The sickness is terrible, au nanuma…We have fallen upon a new age, io e. Infectious disease is spreading among us, io e…71
A Fijian meke (dance/chant), circa 1792, describing the first Pacific-wide pandemic, lila balavu (an unidentified disease), probably imported by HMS Pandora whilesearching for mutineers of HMS Bounty
“Virgin soil epidemic” refers to an infectious disease epidemic introduced into a completely susceptible population. The term seems to have taken hold following an 1875 British parliamentary speech in which Colonial Secretary Lord Carnarvon described an epidemic that had just killed a third of the population of Great Britain’s new Crown Colony, Fiji.[20] and [72]. The Fiji tragedy began innocently. On Jan 12, 1875, HMS Dido sailed into Fiji with its former king and queen, two of their sons, and a retinue of about 100 Fijians. As a sign of good will, Queen Victoria had sent them all on a post-cession sightseeing visit to Australia. 13 days into the 19-day return voyage, one of the sons developed fever and rash. The ship surgeon diagnosed measles, then prevalent in Sydney, but as yet unknown in Fiji.
HMS Dido’s yellow quarantine flag was not flying when it sailed into the Fijian port. Arriving British dignitaries compounded the mistake by focusing on a seemingly more pressing problem: while in Sydney a just-married (now measles-infected) son of the king had acquired “a drip” (gonorrhoea) that had to be kept from newspaper reporters. During the ensuing discussions on board, no one seems to have considered the problem of 100 Fijians potentially incubating measles. Nor did anyone notice that boats were bringing impatient passengers to shore, or that most of Fiji’s police force had sailed out to celebrate on the ship.
Worse yet, the mountain cannibal chiefs had been threatening to revolt against cession and, while the king was away, his brother, Ratu Savanaca Naulivou, had arranged a national meeting, unprecedented in size, at which the chiefs might be persuaded to join the government. On Jan 25, 13 days (about one measles incubation period) after HMS Dido‘s return, the meeting was attended by 69 chiefs, 800 family and guests, and the 143-man police force, many already developing fevers and rashes.20
The attendees returned to their homes throughout Fiji, seeding a widespread and deadly measles epidemic that killed a third of Fiji’s population, an estimated 40 000 people,20 and was exported to other Pacific islands in a regional pandemic. To reduce fever, terrified Fijians lay down in the ocean and in streams and wrapped small children in wet grass. Trying to stop epidemic spread, those who were unafflicted burned the villages of ill, with friends and family still trapped inside their dwellings. Corpses lay everywhere in the open, scavenged by dogs and wild pigs. The stench of destroyed villages was notable a mile away. Ratu Savanaca succumbed and his body was thrown into a communal pit. The mountain chiefs revolted, killing and sometimes eating British subjects. They were defeated by British forces in The Little War, and their leaders were hung. 20 years later, when Queen Victoria commissioned a study of Fiji’s population decrease, the emerging measles epidemic was barely mentioned;73 thus Fijian “racial degeneracy” remained the eugenics-inspired explanation for the decrease. The cautionary tale of Fiji’s quintessential virgin soil epidemic remained in textbooks for another century, but the determinants of its emergence were largely ignored.
Emerging pandemic mysteries: the Spanish influenza, 1918–19
It is quite probable that influenza will continue to be prevalent…all over the world for some years to come…May we hope that etiological and epidemiological work…will furnish us with more competent methods for prevention and delimation before the world is visited by another pandemic.
Hans Zinsser, 1922 74
The 1918 influenza pandemic, arguably the deadliest emerging pandemic to date, is believed to have killed 50–100 million people within the first 2 years of its appearance (figure 2).[75] and [76] The pandemic defied many of the expectations surrounding influenza, appearing in three waves within a 9–12-month period, providing conflicting evidence with regard to wave-to-wave protective immunity, and causing a higher than expected death rate across a wide age-range. The pandemic was also notable for a predominance of severe and fatal disease in healthy young adults.[77], [78] and [79] The 1918 influenza pandemic was one of the first infectious disease outbreaks in which microbial co-infections were appreciated and studied scientifically,80 a phenomenon important to understanding AIDS seven decades later.
The pandemic’s unresolved mysteries have haunted successive generations,78 providing a lesson about the unpredictability of emerging infections. More recently, it has served as a benchmark for pandemic “worst-case scenario” planning.81 While work on the potential for emerging influenza pandemics continues, other worrisome diseases continue to emerge—eg, SARS, appearing in 2003 and caused by a virus genetically unrelated to influenza that nonetheless mimics it in its means of spread and its threat to public-health control.
Out of Africa: the HIV/AIDS pandemic, from 1981
[The Spanish] influenza was the last of the classic pestilences…AIDS…is the first of the postmodern plagues.
Mirko D Grmek, 1989 82
27 years after its recognition, more than 25 million people have died of AIDS; another 33 million remain infected with HIV, most not yet receiving life-prolonging antiviral therapy.[22], [82] and [83] To date, no one with HIV infection has been cured. Within a few years the HIV/AIDS pandemic may surpass the total lethality of the 1918 influenza pandemic to achieve the dubious distinction of being the deadliest emerging disease in recorded history. Like the Black Death of the 14th century, AIDS has rapidly caused high mortality and led to profound social and economic disruption that creates conditions supporting further spread.82
Since at least the time of Thucydides, one of the first human responses to disease emergence has been to question its origin. When identified in 1981, AIDS seemed to be a completely new disease. Its early recognition in a peripatetic airline steward suggested (only partly correctly) that its emergence might have resulted from increased human movement resulting from ever decreasing travel times to multiple destinations.84 Moreover, rapid spread via blood and body fluids within risk groups most capable of supporting such transmission (men who have sex with men, injection drug users, and transfusion recipients) seemed to support recent emergence. Surprisingly, however, phylogenetic analyses of the virus suggest that HIV infection emerged as a human disease around 1900,85 and by complex pathways. Multiple jumps, into chimpanzees, of simian immunodeficiency virus (SIV) strains restricted to different primate species,86 were apparently followed by SIV genetic recombinations that produced new chimpanzee-adapted SIVs. It is likely that three of these recombinants then transferred independently into human beings, leading to parallel HIV emergences now represented by different HIV clades.85
About 1% of Europeans are resistant to HIV because of a homozygous 32 bp deletion in the gene encoding for the major co-receptor for HIV, the human CCR5 cell-surface molecule. This protective allele seems to have appeared in the distant past and to have been positively selected for over many centuries. Geneticists and demographers generally agree that the driving force behind this allele’s frequency was probably an infectious disease, although historical, mathematical, and archeological evidence is incomplete for the two chief contenders, smallpox and plague.87
Discussion
Emerging infections have for millennia threatened the survival of human societies who share ecosystems with rapidly evolving microbial organisms and their non-human hosts, vectors, and reservoirs. Man’s struggle against emerging infections has been a fundamental determinant of the existence and evolution of the human species. The possibility that individual and population resistance to emerging pandemic diseases results from gene selection by previous emerging diseases underscores the complex nature of the human–microbial interaction.87
The ten historical emerging epidemics/epizootics examined here were selected non-systematically from among many newly emerging and re-emerging diseases recorded over several millennia. We attempted to provide examples that were not only well documented but also reflective of the range and complexity of individual and interacting risk determinants. Although these examples cannot be considered representative of all of the many hundreds of historically documented emerging diseases, inclusion of those widely considered among the most important, such as the Black Death, Spanish influenza, and AIDS, serves to highlight determinants relevant at least to emerging diseases with pronounced impact on human beings.
Comparison of historical determinants of emergence and those associated with modern emerging diseases suggests that they are largely the same.5 Prominent among these are determinants reflecting human mobility such as demographics/behaviour and trade/commerce, host susceptibility factors, and poverty/social inequality (panel and table).5 For example, although separated by millennia, the spread along trade routes of newly emerging diseases in ancient Greece (the Plague of Athens), in medieval Europe (the Black Death), and across the African continent (AIDS), are similar in their dependence on human modes and patterns of movement. Other less universal determinants of historical emergence have nonetheless appeared repeatedly in association with varying other cofactors—eg, the role of technology and industry in the re-emergence of diseases such as dengue and chikungunya, spread by the Aedes aegypti mosquito, which oviposits preferentially in modern products such as discarded tyres and tin cans.
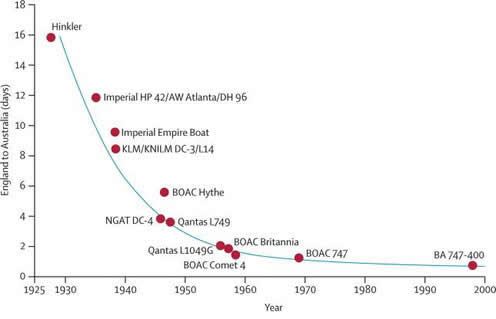
If there is anything new in disease emergence today, it seems to be less in the nature of specific determinants of emergence than in the greater complexity of modern existence, which in turn leads to increased opportunities for convergence of interactive risk determinants and greater speeds with which emergence can occur and escape control measures. As long ago as 1918, in the era before commercial air travel, the second wave of the influenza pandemic appeared in different regions of the world almost simultaneously. In 2008, air travel can bring a new disease from any major city to any other in a day (figure 3), while complex trade and distribution systems can spread even a non-contagious infectious disease quickly beyond the reach of local public-health containment, as exemplified by a US national ice cream-associated epidemic of salmonella in 199488 and a spinach-associated epidemic of Escherichia coli O157:H7 in 2006.89 Moreover, recent recognition of the role of weather/climate, climate cycles (eg, the El Niño southern oscillation), and global warming in the emergence of diseases such as dengue and malaria suggests that risk determinant complexities and interactions may increase.6
Figure 3. Average travel time between England and Australia, 1925–2000
As travel times between distant places have decreased markedly in the past nine decades, the potential for human importation of diseases, especially those with long incubation periods—even if they are easily identifiable—is greatly increased, thereby increasing the potential for re-emergence of epidemic diseases via spread into new geographic areas. The serial generation time of measles is approximately 14 days. Adapted from Cliff et al, 2000,84 by permission of Oxford University Press.
Although this article has emphasised emerging pandemic diseases, emerging diseases with more limited (non-pandemic) spread, such as hantavirus pulmonary syndrome and Lyme disease, have also presented substantial public-health challenges. In other cases, disease emergence may occur so insidiously that recognition is difficult (eg, tuberculosis during the Renaissance [14–17th centuries]). In recent times we have seen an extraordinary growth in re-emergence of infectious diseases, a phenomenon that apparently began in the Age of Discovery (15–18th centuries), when microbes came into contact with new human populations, and which is still accelerating as population growth and travel provide better avenues of escape for microbes entrenched in complex localised ecosystems (eg, geographic extension of arboviral diseases such as dengue, Japanese encephalitis, West Nile virus, and chikungunya infections).
Conclusion
Well-understood determinants of modern disease emergence, typically acting in concert, have been associated throughout recorded history with the emergence of major diseases. These determinants have been similar in their explosiveness, impact, and elicitation of public-health control responses. Whether the nature and pattern of these determinants are changing or will change in the future remains speculative. That most of the historical emerging diseases we examined were associated with unique patterns of common determinants suggests to us that an increasingly complex modern world will probably provide increasing opportunities for disease emergence. For centuries a fundamental challenge to the existence and well-being of societies—as reflected by scientific attention, as well as in art, religion, and culture—emerging infections remain among the principal challenges to human survival.
Conflicts of interest
We declare that we have no conflicts of interest.
Acknowledgments
We thank Jeffery K Taubenberger and the staff of the History of Medicine Division and the Public Services Division, National Library of Medicine, National Institutes of Health, for assistance with the manuscript and for obtaining some of the cited references.
References
- R Virchow, Ueber die neueren Fortschritte in der Pathologie, mit besonderer Beziehung auf öffentliche Gesundsheitspflege und Aetiologie. (Rede, gehalten in der zweiten allgemeinen Sitzung der Deutsches Naturforscher-Versammlung zu Frankfurt a M am 20 September 1867). In: R Virchow, Editor, Gesammelte Abhandlungen aus dem Gebiete der öffentlichen Medicin und der Seuchen-lehre. Erster Band. XI, August Hirschwald, Berlin (1879), pp. 96–107.
- WHO, The world health report 2007—a safer future: global public health in the 21st Century, World Health Organization, Geneva (2007) http://www.who.int/whr/2007/en/index.html (accessed Sept 15, 2008)..
- In: J Lederberg, RE Shope and SC Oaks, Editors, Emerging infections: microbial threats to health in the United States, Institute of Medicine, National Academy Press, Washington, DC (1992).
- DS Stephens, ER Moxon and J Adams et al., Emerging and reemerging infectious diseases: a multidisciplinary perspective, Am J Med Sci315 (1998), pp. 64–75.
- DM Morens, GK Folkers and AS Fauci, The challenge of emerging and re-emerging infectious diseases, Nature430 (2004), pp. 242–249.
- In: MS Smolinski, MA Hamburg and J Lederberg, Editors, Microbial threats to health: emergence, detection, and response, National Academies Press, Washington, DC (2003).
- CR Parrish, EC Holmes and DM Morens et al., Cross-species virus transmission and the emergence of new epidemic diseases, Microbiol Mol Biol Rev72 (2008), pp. 457–470.
- NM Ampel, Plagues—what’s past is present: thoughts on the origin and history of new infectious diseases, Rev Infect Dis13 (1991), pp. 658–665.
- RM Krause, The origin of plagues: old and new, Science257 (1992), pp. 1073–1078.
- 10 N Webster, Letters on yellow fever addressed to Dr William Currie. Supplement to The Bulletin of the History of Medicine. No 9, Johns Hopkins University Press, Baltimore (1947).
- H Haeser, Historische-pathologische Untersuchungen. Als Beiträge zur Geschichte der Volkskrankheiten, Gerhard Fleischer, Dresden (1839–41).
- KF Cheng and PC Leung, What happened in China during the 1918 influenza pandemic?, Int J Infect Dis11 (2007), pp. 360–364.
- Thucydides, The hystory, writtone by Thucidides the Athenyan, of the warre: whiche was betwene the Peloponesians and the Athenyans, William Tylle, London (1550).
- JF Hecker, Der schwarze Tod im vierzehnten Jahrhundert, Friedrich August Herbig, Berlin (1832).
- C Quétel, Le mal de Naples: histoire de la syphilis, Éditions Seghers, Paris (1986).
- AW Crosby, The Columbian exchange: biological and cultural consequences of 1492, Greenwood Press, Westport, Connecticut (1972).
- DM Morens, Characterizing a “new” disease: epizootic and epidemic anthrax, 1769–1780, Am J Public Health93 (2003), pp. 886–893.
- JH Powell, Bring out your dead; the great plague of yellow fever in Philadelphia in 1793, Arno Press, New York (1970).
- L-F Benoiston de Châteauneuf, J-B Chevallier and L Devaux et al., Rapport sur la marche et les effets du choléra-morbus dans Paris et les communes rurales du département de la Seine, Imprimerie Royale, Paris (1834).
- DM Morens, Measles in Fiji, 1875. Thoughts on the history of emerging infectious diseases, Pac Health Dialog5 (1998), pp. 119–128.
- EO Jordan, Epidemic influenza: a survey, American Medical Association, Chicago (1927).
- AS Fauci, HIV and AIDS: 20 years of science, Nat Med9 (2003), pp. 839–843
- DM Morens, Epidemic anthrax in the eighteenth century, the Americas, Emerg Infect Dis8 (2002), pp. 1160–1162. View Record in Scopus | Cited By in Scopus (4)
- de’Mussis G. Manuscript R 262; fos 74r. Library of the University of Wroclaw, Poland, c 1348.
- M Wheelis, Biological warfare at the 1346 siege of Caffa, Emerg Infect Dis 8 (2002), pp. 971–975.
- OJ Benedictow, The territorial origin of plague and of the Black Death. In: OJ Benedictow, Editor, The Black Death, 1346–1353. The Complete History, Boydell Press, Woodbridge, Suffolk (2004), pp. 35–54 Chapter 5.
- H Daumier, Quatrième satire. Souvenirs du choléra-morbus; Notes. In: A-H Fabre, Editor, Némésis médicale illustré. Recueil de satires… Contenant trente vignettes dessinées par M. Daumier, et gravées par la meilleurs artistes, avec un grand nombre de culs-de-lampe, etc. Tome premier, Bureau de la Némésis Médicale, Paris (1841), pp. 63–89 91–94..
- DM Morens and RJ Littman, Epidemiology of the “Plague of Athens”, 430–426 BC, Trans Am Philol Assoc 122 (1992), pp. 271–304.
- W Boghurst, Loimographia: an account of the Great Plague of London in the year 1665, Shaw & Sons, London (1979).
- GC Kohn, The Wordsworth encyclopedia of plague and pestilence, Facts on File, New York (1995).
- JF Hecker, Die Tanzwuth, eine Volkskrankheit im Mittelalter. Nach den Quellen für Aerzte und gebildete Nichtärzte, Enslin, Berlin (1832).
- DM Morens, Mass fainting at medieval rock concerts, N Engl J Med 333 (1995), p. 1361.
- Jacme, J. Regiment de preservació a epidemia o pestilència e mortaldats. Lerida: Council of Lerida, 1348.
- GW Christopher, TJ Cieslak, JA Pavlin and EM Eitzen, Biological warfare. A historical perspective, JAMA 278 (1997), pp. 412–417.
- RL Koenig, The fourth horseman, Public Affairs, New York (2006).
- U von Hutten, De guaiaci medicina et morbo gallico liber unis, Joannis Scheffer, Mogu[n]tiae (Mainz) (1519).
- G de Vigo, Practica in arte chirurgica copiosa… continens novem libros…, Stephanum Guillireti & Herculem Bononiensem socium, Rome (1514).
- Erasmus D. Lingua sive, de linguae usu, atque abusu, Liber unus. 1520.
- Fracastoro G. Syphilis, sive morbus gallicus. Veronæ, 1530.
- G Fracastoro, De sympathia et antipathia rerum liber unus. De contagione et contagiosis morbis et curatione libri iii, heredes Lucaeantonii Iuntae Florentini, Venetiis (1546).
- GF de Oviedo y Valdés, La historia general de las Indias. Con privilegio imperial… Fin de la primera parte dela general y naturel hystoria delas indias yslas y tierra firme del mar oceano, Iuam Cromberger, Sevilla (1535).
- R Díaz de Isla, Tractado c[on]tra el mal serpentino: que vulgarmente en España es llamado bubas q fue ordenado en el Ospital de Todos Los Santos de Lisbona, D de Robertis, Sevilla (1539).
- S Sontag, AIDS and its metaphors, Farrar, Straus & Giroux, New York (1989).
- F Hernández Arana and F Díaz, [Translated by Brinton DC]. The annals of the Cakchiquels, University of Pennsylvania, Philadelphia (1885).
- B de Sahagún, Historia general de las cosas de Nueva España. [“Florentine Codex”], Alejandro Valdes, Mexico City (1829).
- SF Cook and W Borah, The Indian population of central Mexico, 1531–1610, University of California Press, Berkeley (1960).
- W Borah and SF Cook, Additional remarks. In: SF Cook and W Borah, Editors, The Aboriginal population of central Mexico on the eve of the Spanish conquest VII, University of California Press, Berkeley (1963), pp. 89–92.
- F Fenner, Smallpox: emergence, global spread, eradication, Hist Philos Life Sci 15 (1993), pp. 397–420.
- FF Cartwright and MD Biddiss, The impact of infectious diseases. In: FF Cartwright and MD Biddiss, Editors, Disease and history, Rupert Hart-Davies, London (1972), pp. 113–136 Chapter 5..
- S Riedel, Biological warfare and bioterrorism: a historical review, Proc (Bayl Univ Med Cent) 17 (2004), pp. 400–406.
- F Vicq-d’Azyr, Contenant les moyens curatifs. In: F Vicq-d’Azyr, Editor, Exposé des moyens curatifs & préservatifs qui peuvent être employés contre les maladies pestilentielles des bêtes à cornes. Première partie, Mérigot l’aîné, Paris (1776).
- B Ramazzini, De contagiosa epidemia, quæ in Patavino Agro, & tota ferè Veneta. Ditione in Boves irrepsit. Dissertatio habita in Pavatino Lyceo A’ Bernardino Ramazzini, Practicæ Medicinæ Professore Primario. Die IX. Novembris MDCCXI, Jo[annis] Baptistæ Conzatti, Patavii (1712).
- Z Boylston, An historical acount of the small-pox inoculated in new England, upon all sorts of persons, whites, blacks, and of all ages and constitutions, S Chandler, London (1726).
- J-P Desaive, J-P Goubert, E Le Roy Ladurie, J Meyer, O Muller and J-P Peter, Médecins, climat et épidémies à la fin du XVIIIe siècle, Mouton, Paris (1972).
- DB Jernigan, PL Raghunathan and BP Bell et al., Investigation of bioterrorism-related anthrax, United States, 2001: epidemiologic findings, Emerg Infect Dis 8 (2002), pp. 1019–1028.
- M-P Justin, Livre troisième. In: M-P Justin, Editor, Histoire politique et statistique de l’ Île d’Hayti, Saint-Domingue, écrite sur les documents officiels et des notes communiquées par Sir James Barskett, Brière, Paris (1826), pp. 109–125.
- M Meselson, J Guillemin and M Hugh-Jones et al., The Sverdlovsk anthrax outbreak of 1979, Science 266 (1994), pp. 1202–1208.
- JP Krieg, The American plague. In: JP Krieg, Editor, Epidemics in the modern world, Twayne Publishers, New York (1992), pp. 41–67 Chapter 3
- L Spalding, A dissertation on the bilious malignant fever which prevailed in the country adjacent to Dartmouth College, in the summer of 1798, The Medical Repository 3 (1805), pp. 5–13.
- R Ketcham, Wartime president. In: R Ketcham, Editor, James Madison: a biography, University of Virginia Press, Charlottesville (1990), pp. 534–572 Chapter 20..
- J Murphy, Confusion, distress, and utter desolation. In: JP Murphy, Editor, An American plague, Clarion Books, New York (2003), pp. 35–45 Chapter 4..
- A Jones, R Allen, M Clarkson and WW Woodward, A narrative of the proceedings of the black people, during the late awful calamity in Philadelphia, in the year 1793: and a refutation of some censures, thrown upon them in some late publications, William W Woodward, Philadelphia (1794).
- R Allen, The life, experience, and gospel labours of the Rt Rev Richard Allen to which is annexed the rise and progress of the African methodist episcopal church in the United States of America. Containing a narrative of the yellow fever in the year of Our Lord 1793: with an address to the people of colour in the United States, Martin & Boden, Philadelphia (1833).
- W Linn, A discourse delivered on the 26th day of November, 1795; Being the day recommended by the Governor of the state of New-York to be observed as a day of thanksgiving and prayer, on account of the removal of an epidemic fever, and for other national bessings, T and J Swords, New York (1795).
- B Furman, John Adams—federal health insurance for seamen exposed to epidemics 1790–1800. In: B Furman, Editor, A profile of the United States Public Health Service, 1798–1948, US Government Printing Office, Washington, DC (1973), pp. 1–21 Chapter 1..
- RJ Kahn and PG Kahn, The Medical Repository—the first US medical journal (1797–1824), N Engl J Med 337 (1997), pp. 1926–1930.
- N Webster, A brief history of epidemic and pestilential diseases with the principal phenomena of the physical world, which precede and accompany them, and observations deduced from the facts stated, Hudson & Goodwin, Hartford, Connecticut (1799).
- A Hirsch, Handbuch der historisch-geographischen Pathologie, Ferdinand Enke, Stuttgart (1881–86).
- L-F Benoiston de Châteauneuf, Précautions prises par l’administration avant l’invasion du choléra. In: L-F Benoiston de Châteauneuf, J-B Chevallier and L Devaux et al., Editors, Rapport sur la marche et les effets du choléra-morbus dans Paris et les communes rurales du département de la Seine. Chapitre premier, Imprimerie royale, Paris (1834), pp. 11–19.
- DM Morens, “Epidemiology is born”: cholera in Paris, 1832. In: RH Bernier and JL St Pierre, Editors, Epidemiology wit & wisdom, The Epidemiology Monitor Press, Roswell, Georgia (2000), pp. 98–101.
- I Motonicocoka, The story of the lila balavu (wasting sickness) and of the cocadra (dysentery); and the meke relating to these events. In: BG Corney, J Stewart and BH Thomson, Editors, Report of the commission appointed to inquire into the decrease of the native population, with appendices, John Edward March, Suva, Fiji (1896) [translator]. Appendix I..
- H Molyneux, 50 000 natives die at Fiji, The Times (June 9, 1875), p. 7 (London).
- BG Corney, J Stewart and BH Thomson, Report of the commission appointed to inquire into the decrease of the native population, with appendices, John Edward March, Suva, Fiji (1896).
- H Zinsser, The etiology and epidemiology of influenza, Medicine 1 (1922), pp. 213–309.
- NP Johnson and J Mueller, Updating the accounts: global mortality of the 1918–1920 “Spanish” influenza pandemic, Bull Hist Med 76 (2002), pp. 105–115.
- CJ Murray, AD Lopez, B Chin, D Feehan and KH Hill, Estimation of potential global pandemic influenza mortality on the basis of vital registry data from the 1918–20 pandemic: a quantitative analysis, Lancet 368 (2006), pp. 2211–2218.
- H French, The clinical features of the influenza epidemic of 1918–19, Great Britain. Ministry of Health. Reports on public health and medical subjects. No 4. Report on the pandemic of influenza, 1918–19, His Majesty’s Stationery Office, London (1920), pp. 66–109 Chapter III..
- DM Morens and AS Fauci, The 1918 influenza pandemic: insights for the 21st century, J Infect Dis 195 (2007), pp. 1018–1028.
- JK Taubenberger, JV Hultin and DM Morens, Discovery and characterization of the 1918 pandemic influenza virus in historical context, Antivir Ther 12 (2007), pp. 581–591.
- DM Morens, JK Taubenberger and AS Fauci, Predominant role of bacterial pneumonia as a cause of death in pandemic influenza: implications for pandemic influenza preparedness, J Infect Dis 198 (2008), pp. 962–970.
- United States Department of Health and Human Services, HHS pandemic influenza plan, US Government Printing Office, Washington, DC (2005).
- MD Grmek, Histoire du sida: début et origine d’une pandémie actuelle, Éditions Payot, Paris (1989).
- UNAIDS, AIDS epidemic update: December 2006, UNAIDS, Geneva (2006) http://www.unaids.org/en/KnowledgeCentre/HIVData/EpiUpdate/EpiUpdArchive/2006/Default.asp (accessed Sept 15, 2008).
- AD Cliff, P Haggett and M Smallman-Raynor, The changing accessibility of islands. In: AD Cliff, P Haggett and M Smallman-Raynor, Editors, Island epidemics, Oxford University Press, Oxford (2000), pp. 165–236.
- M Worobey, M Gemmel and DE Teuwen et al., Direct evidence of extensive diversity of HIV-1 in Kinshasa by 1960, Nature 445 (2008), pp. 661–664.
- E Bailes, F Gao and F Bibollet-Ruche et al., Hybrid origin of SIV in chimpanzees, Science 300 (2003), p. 1713.
- J Novembre, AP Galvani and M Slatkin, The geographic spread of the ΔCCR5 32 HIV-resistance allele, PLoS Biol 3 (2005), pp. 1954–1962.
- TW Hennessy, CW Hedberg and L Slutsker et al., A national outbreak of Salmonella enteritidis infections from ice cream, N Engl J Med 334 (1996), pp. 1281–1286.
- M Cooley, D Carychao and L Crawford-Miksza et al., Incidence and tracking of Eschericia coli O157:H7 in a major produce production region of California, PLoS One 11 (2007), pp. 1–16.
Correspondence to: Dr David M Morens, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Building 31, Room 7A10, 31 Center Drive, Bethesda, MD 20892, USA
Highlighted in Frontispiece Winter 2009- Volume 1, Issue 2
Winter 2009 | Sections | Infectious Diseases


Leave a Reply