For much of medical history, pernicious anemia was a baffling and invariably fatal disorder. Characterized by profound weakness, pallor, glossitis, and progressive neurologic decline, it carried an ominous reputation. Its name—“pernicious”—reflected the despair of physicians who could offer no remedy.
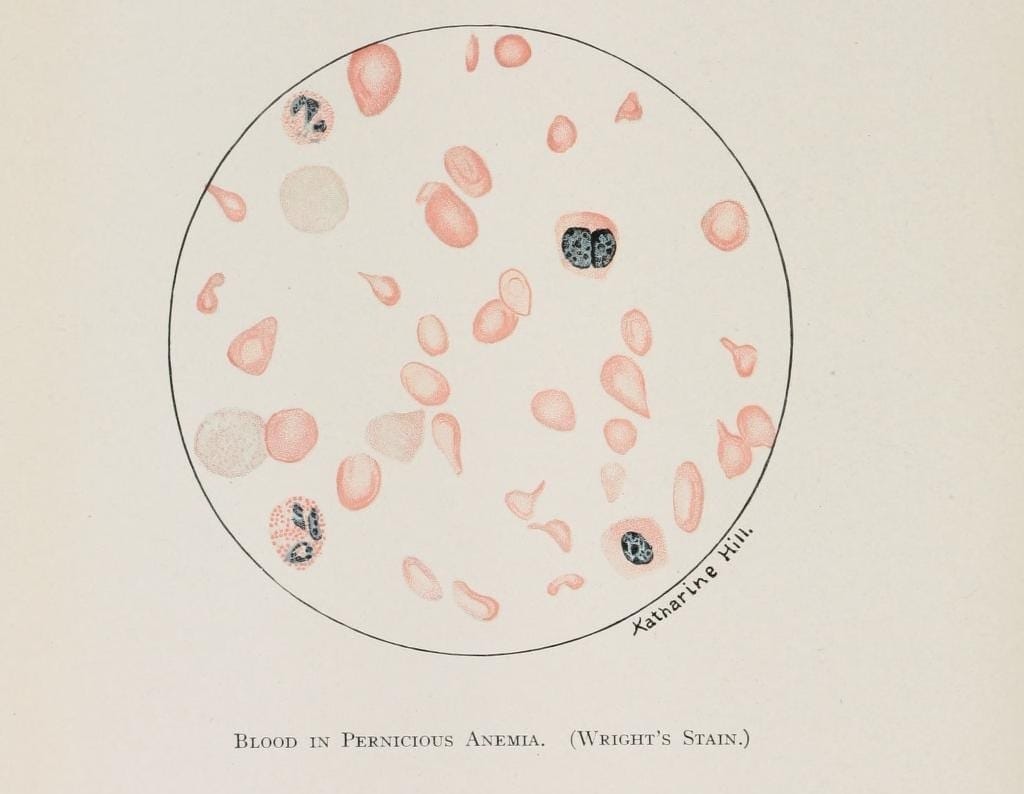
The symptoms of this disease developed gradually and were fatigue, weakness, pallor, and shortness of breath. Neurological symptoms often came later, such as numbness or tingling in the limbs, trouble walking, and, in severe cases, confusion or dementia. Examination of a blood film would reveal large red blood cells (macrocytic or megaloblastic anemia) and low serum vitamin B12 levels. Other tests would reveal elevated methylmalonic acid levels, antibodies against intrinsic factor or parietal cells, absorption problems, or atrophic gastritis.
Pernicious anemia is more common in people of Northern European descent and typically occurs in people over 60 years of age, though it can be found at any age. People with other autoimmune disorders such as type 1 diabetes, vitiligo, or thyroid disease have increased susceptibility. The condition may run in families.
The first clear clinical description of the condition is generally attributed to the English physician Thomas Addison. In 1849, and more fully in 1855 in On the Constitutional and Local Effects of Disease of the Suprarenal Capsules, Addison described patients with anemia and others with bronzed skin due to adrenal insufficiency. He did not understand their cause, but he recognized their relentless and fatal nature.
In the nineteenth part of the century, the German physician Anton Biermer provided more detailed accounts and introduced the term “progressive pernicious anemia.” Biermer emphasized the severe macrocytic anemia seen on blood examination and the progressive, often fatal outcome. By this time, advances in microscopy had allowed physicians to observe abnormally large red blood cells—what we now call megaloblasts—although their significance was not yet understood. The bone marrow, when examined, showed striking hypercellularity with ineffective erythropoiesis, but the underlying defect remained obscure. A crucial clue, however, emerged from pathology: many patients had atrophic gastritis, with thinning of the gastric mucosa. Yet the connection between the stomach and the bone marrow was not immediately apparent.
The decisive breakthrough came in the 1920s. George Whipple, studying anemia in dogs, discovered that feeding them liver accelerated recovery from blood loss. In 1926 George Minot and William Murphy applied this observation to patients with pernicious anemia and reported dramatic improvements in patients who consumed large quantities of raw liver daily. This was nothing short of revolutionary. Bedridden patients regained strength, blood counts rose, and neurologic symptoms sometimes improved. In 1934 Whipple, Minot, and Murphy were awarded the Nobel Prize in Physiology or Medicine.
Yet liver was a cumbersome treatment—patients often had to eat half a pound or more daily. The next advance occurred in 1948 when two research groups independently identified vitamin B12 (cobalamin) as the crucial anti-pernicious anemia substance. This discovery transformed therapy; instead of consuming large amounts of liver, patients could receive small, precise doses of vitamin B12 by injection. But one puzzle remained: why could patients with pernicious anemia not simply absorb vitamin B12 from food? The answer lay in the physiology of the stomach. Researchers revealed that patients with pernicious anemia lacked “intrinsic factor,” a glycoprotein produced by parietal cells of the gastric mucosa. Intrinsic factor binds vitamin B12 and facilitates its absorption in the terminal ileum. Without it, even a diet rich in B12 cannot prevent deficiency. The atrophic gastritis observed decades earlier was not incidental; it was central. Pernicious anemia is now understood as an autoimmune disease in which antibodies target the intrinsic factor and the gastric parietal cells, leading to impaired B12 absorption.
