Beatriz GT Pogo
In 1964 I joined the laboratory of Cell Biology of Vincent G. Allfrey and Alfred E. Mirsky at the Rockefeller University as a post-doctoral fellow supported by a Damon Runyon International Fellowship.
Early that year Rosemary Faulkner had finished her Ph.D. thesis under Dr. Vincent Allfrey on histone acetylation in isolated thymus nuclei. Allfrey had observed a positive correlation between histone acetylation and RNA synthesis in isolated nuclear fractions, but the important question was if this phenomenon occurred in vivo.1 The hypothesis at that time was that histone acetylation would change the fine structure of chromatin, as a neutralization mechanism, thus derepressing DNA to serve as a template for RNA synthesis. I discussed with Allfrey what cell system was to be used. I had previously worked on (A) gene activation using induction of chloroplast differentiation by light, and also (B) the stimulation of mitosis in peripheral blood lymphocytes by mitogens. I suggested to Allfrey that we use the latter system to look for gene activation which in those days meant RNA synthesis, and simultaneously for histone acetylation. He was an intelligent, enthusiastic and knowledgeable investigator. He liked the idea – and I proceeded.
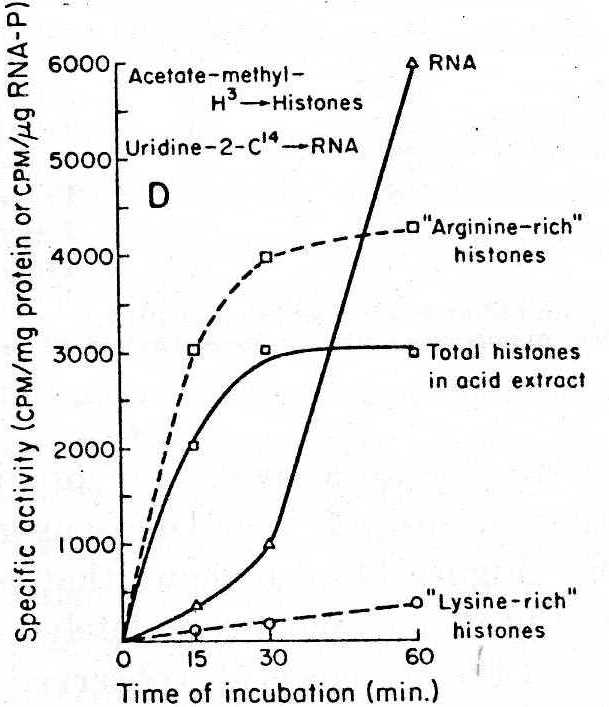
In New York at that time, human blood was easily purchased from seedy commercial blood banks before the New York Blood Center under better regulations was established. We separated mononuclear cells from other white blood cells and cultured these isolated mononuclear cells in the presence of phytohemoagglutinin (PHA), a plant mitogen. A burst of RNA synthesis took place after a few hours of incubation and mitosis occurred after 48-72 hours of culture. Histone acetylation was measured by 14C-acetate incorporation into the histones separated by electrophoresis, and RNA synthesis was independently measured by 14C-uridine uptake into isolated RNA. Both processes were shown to be stimulated after a short exposure to PHA. We also demonstrated that the 14C-acetate was incorporated as an acetyl group and was not converted to an amino acid, and that the so-called “arginine-rich histones” were most effectively acetylated. By pulse-labelling experiments we also showed that histone acetylation preceded RNA synthesis. These results were very exciting. The human blood provided a good start, but to do more biochemical work, it was necessary to have more cells with which to work. Pooling mononuclear preparations from different donors was out of the question because it meant introducing another uncontrolled factor. A postdoctoral fellow, whose name unhappily I do not recall, told me she was using equine white cells for her immunological work. The Rockefeller Foundation at Rockefeller University had a stable with a well-kept old police horse which was used to provide the laboratories with horse serum, then a common reagent used in tissue culture. That old horse gave us sufficient blood cells to allow the biochemical characterization of the system.
In 1966 we published the first paper on histone acetylation and RNA synthesis in equine lymphocytes stimulated by PHA.2 These observations were then extended to other system like rat liver regeneration.3,4 We also showed by autoradiography with Virginia Littau that during RNA synthesis in chromosomes of insect salivary glands there was acetylation of histones as well.5 In addition to investigating acetylation, Allfrey analyzed the phosphorylation and methylation of histones, as two of his students completed their doctoral dissertations on the subject. 6,7
In 1968, Gershey et al, published the identification of E-N-acetyl lysine in the f2a1 histone.8 Although we then realized the importance of changes in chromatin structure and gene de-repression, we did not think of the profound significance these early observations would have on future drug design and treatment.9 Nor did we realize that the field of Epigenetics had been born. I left Allfrey’s lab with great regret in a moment of crisis, his separation from Alfred Mirsky after many years of collaboration. I also realized that the Rockefeller University was not then a likely place for a woman to make a career. The New York Public Health Research Institute – called, at that time, “little Rockefeller” because of the presence of so many graduates, postdocs, and faculty from Rockefeller University – offered an excellent scientific environment and opportunities. I left but I always missed those three years (and nine papers) of intense work in Allfrey’s lab at the Rockefeller University and the exciting field of histone post-translation modifications. It was the unwritten but widely understood rule that after one left, one could not work on the subject of the laboratory where one had been trained.
Vincent Allfrey did continue to work on the subject and made important contributions to the field. Unfortunately, he died in 2002 before epigenetics became what it is today. The last time I saw him was at a conference in the Caspary Auditorium of Rockefeller University. He told me that he was writing a review and had looked into my old laboratory books and how good and clear were my protocols. It was very rewarding to be praised by my mentor decades later. A few months after, he died, but his legacy lives on.10
References
- Allfrey VG, Faulkner R, and Mirsky AE. Acetylation and methylation of histones and their possible role in the regulation of RNA synthesis. Proc Natl Acad Sci (USA) 51:786-812, 1964.
- Pogo BGT, Allfrey VG, and Mirsky AE. RNA synthesis and histone acetylation during the course of gene activation in lymphocytes. Proc Natl Acad (USA) 55:805-812, 1966.
- Pogo BGT, Pogo AO, Allfrey VG, and Mirsky AE. Changing patterns of histone acetylation and RNA synthesis in regeneration of the liver. Proc Natl Acad Sci (USA) 59:1337-1344, 1968.
- Pogo BGT, Pogo AO, and Allfrey VG. Histone acetylation and RNA synthesis in rat liver regeneration. Genetics Supplement 61(1):373-379, 1969.
- Allfrey VG, Pogo BGT, Littau VC, Gershey EL, and Mirsky AE. Histone acetylation in insect chromosomes. Science 159:314-316, 1968.
- Kleinsmith LJ, Allfrey VG, and Mirsky AE. Phosphoprotein metabolism in isolated lymphocytes nuclei. Proc Natl Acad Sci (USA) 55:1182-1189, 1966.
- Tidwell T, Allfrey VG, and Mirsky AE. The methylation of histones during liver regeneration. J Biol Chem 243:707-715, 1968.
- Gershey EL, Vidali G, and Allfrey VG. Chemical studies of histone acetylation – the occurrence of E-N-acetylysine in the f2a1 histone. J Biol Chem 243:5018-5022, 1968.
- Li Z and Zhu W-G. Targeting histone deacetylases for cancer therapy: From molecular mechanisms to clinical implications. Int J Biol Sci 10:4, 2014.
- Gottesfeld J and Mukhopadhyay R. Vincent Allfrey’s work on histone acetylation. J Biol Chem 287:2270-2271, 2012.
Acknowledgement:
I would like to thank Dr. James F. Holland of the Mount Sinai School of Medicine for not only suggesting that I write this essay but who also helped with editing.
BEATRIZ GT POGO
Highlighted in Frontispiece Winter 2016 – Volume 8, Issue 1

Leave a Reply